Key Insights
The Electronic Trial Master File (eTMF) Systems market is poised for significant expansion, projecting a robust market size of $1.36 billion in 2025 and is expected to grow at a compelling CAGR of 12.8% during the forecast period of 2025-2033. This substantial growth is primarily fueled by the increasing complexity of clinical trials, stringent regulatory requirements globally, and the growing need for efficient, centralized document management. The adoption of eTMF systems is critical for pharmaceutical companies, biotech firms, and contract research organizations (CROs) to streamline trial processes, enhance data integrity, and ensure compliance with Good Clinical Practice (GCP) guidelines. Key drivers include the escalating volume of clinical trial data, the drive towards decentralized clinical trials (DCTs) which necessitate robust remote access and collaboration features, and the continuous advancements in cloud-based eTMF solutions offering greater scalability and accessibility. The market's dynamism is further evidenced by the ongoing innovation in features such as artificial intelligence (AI) for automated document classification and quality control, and enhanced audit trail capabilities.
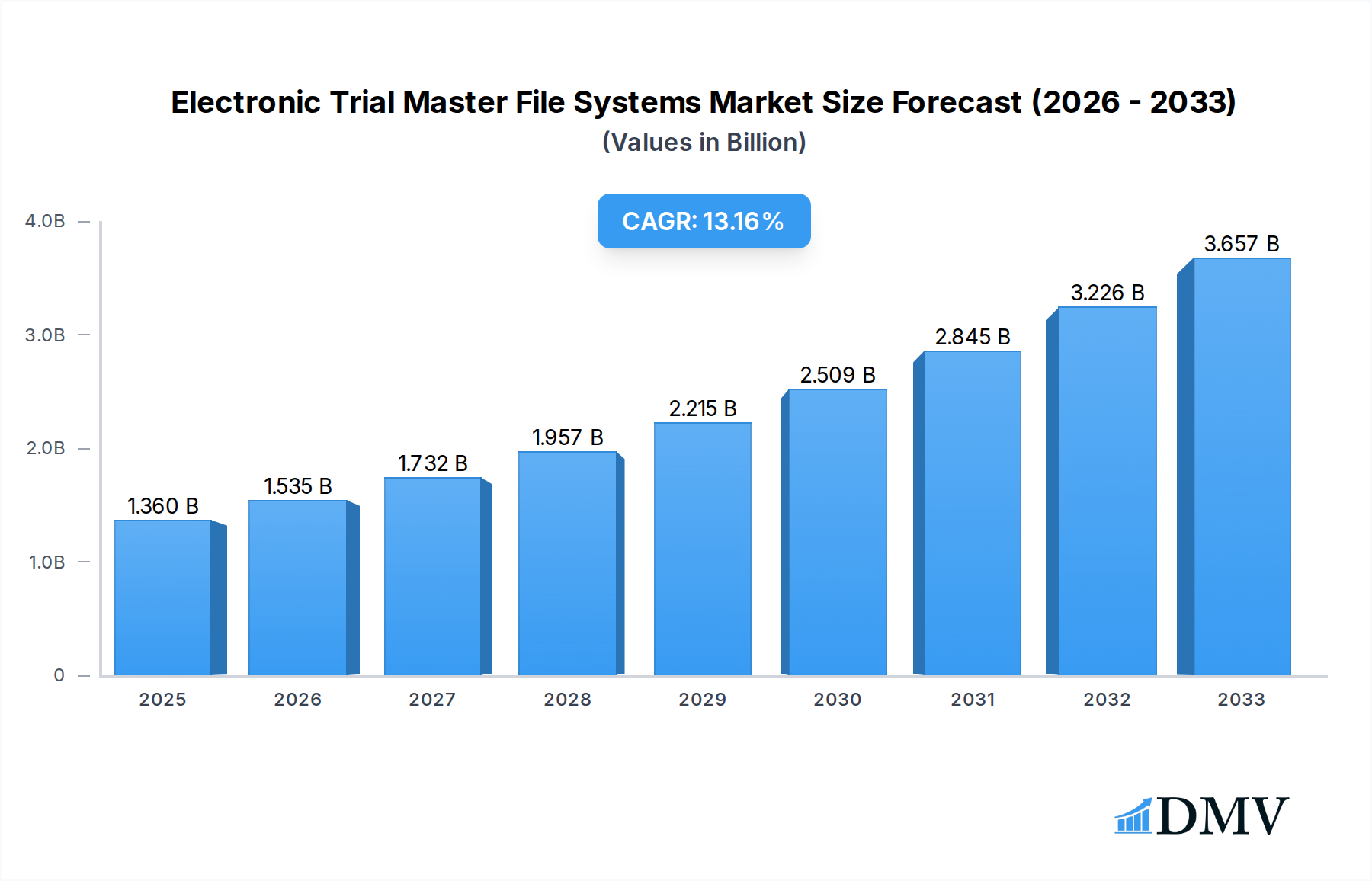
Electronic Trial Master File Systems Market Size (In Billion)

The eTMF Systems market is segmented by application into Hospitals, Clinics, and Ambulatory Care Centers, with Hospitals anticipated to hold a dominant share due to the higher volume and complexity of trials conducted. On-premise and cloud-based deployment models cater to diverse organizational needs, with cloud solutions gaining traction due to their cost-effectiveness, flexibility, and ease of implementation. Major regions like North America and Europe are leading the adoption of eTMF systems, driven by advanced healthcare infrastructure and a strong regulatory framework. However, the Asia Pacific region is expected to witness the fastest growth due to increasing investments in R&D, a burgeoning CRO sector, and a rising number of clinical trials being conducted. Despite the positive outlook, challenges such as the initial cost of implementation, data security concerns, and the need for extensive training for personnel can pose restraints. Nonetheless, the overwhelming benefits of improved operational efficiency, reduced timelines, enhanced collaboration, and superior regulatory compliance are propelling the eTMF market forward, making it an indispensable tool in modern clinical research.
Electronic Trial Master File Systems Company Market Share

Here's the SEO-optimized and insightful report description for Electronic Trial Master File Systems:
Electronic Trial Master File Systems Market Composition & Trends
This comprehensive report dissects the global Electronic Trial Master File (eTMF) systems market, a critical component in modern clinical trial management. We evaluate the market concentration, revealing dominant players like Veeva Systems, Oracle, Aurea Software, MasterControl, Inc., Ennov, Wingspan, Covance Inc, TransPerfect, Phlexglobal, SureClinical Inc, and ePharmaSolutions. Our analysis delves into innovation catalysts, including the increasing adoption of cloud-based eTMF solutions and advancements in AI-driven data management. The report meticulously examines the evolving regulatory landscapes, such as the impact of ICH GCP E6(R2) guidelines, and the growing demand for eTMF software that ensures compliance and data integrity. We explore substitute products and their limitations, underscoring the unique advantages of dedicated eTMF platforms. End-user profiles are meticulously detailed, encompassing hospitals, clinics, and ambulatory care centers, highlighting their specific eTMF requirements and adoption patterns across on-premise and cloud deployment types. Furthermore, the report quantifies Merger & Acquisition (M&A) activities, with historical deal values estimated in the billions, showcasing industry consolidation and strategic partnerships driving market expansion. The eTMF market share distribution reveals a dynamic competitive environment with significant investments pouring into eTMF solutions for clinical research organizations (CROs) and pharmaceutical companies.
- Market Concentration: Highly fragmented with emerging consolidation trends.
- Innovation Catalysts: AI in data review, enhanced interoperability, and advanced analytics.
- Regulatory Impact: Strict adherence to FDA, EMA, and other global regulatory body requirements.
- Substitute Products: Traditional paper-based TMFs and generic document management systems.
- End-User Adoption: Steady growth across all healthcare segments for digitized trial documentation.
- M&A Activities: Significant investments in acquiring innovative eTMF technologies and market reach.
Electronic Trial Master File Systems Industry Evolution
The Electronic Trial Master File (eTMF) systems industry has undergone a remarkable transformation, evolving from nascent digital solutions to sophisticated platforms integral to clinical trial execution. Over the study period of 2019–2033, the market has witnessed exponential growth, driven by the imperative for greater efficiency, enhanced data security, and stringent regulatory compliance. The base year of 2025 represents a pivotal point, with the estimated year of 2025 and the forecast period of 2025–2033 projecting sustained and accelerated adoption. Historical data from the historical period of 2019–2024 clearly illustrates this upward trajectory. Technological advancements have been the cornerstone of this evolution. Initially, eTMF systems focused on digitizing existing paper processes, offering basic document storage and retrieval. However, the industry has rapidly advanced to incorporate features like real-time collaboration, automated quality control checks, advanced audit trails, and seamless integration with other clinical trial management software (CTMS), EDC, and ePRO systems. This technological sophistication is crucial for managing the ever-increasing volume and complexity of clinical trial data.
Shifting consumer demands, particularly from pharmaceutical companies and contract research organizations (CROs), have played a significant role. The pressure to expedite drug development timelines, reduce operational costs, and improve the overall quality of clinical trials has fueled the demand for robust and intuitive eTMF solutions. Stakeholders are actively seeking platforms that can streamline the TMF creation and maintenance process, reduce the risk of errors, and provide real-time visibility into trial progress and compliance. The adoption metrics reflect this demand, with an estimated growth rate of XX% annually during the forecast period. Early adopters of cloud-based eTMF systems have consistently reported higher levels of operational efficiency and faster audit readiness. The market has responded by prioritizing the development of user-friendly interfaces, scalable architectures, and comprehensive security features to meet the diverse needs of global clinical research. The industry's evolution is characterized by a relentless pursuit of innovation, aiming to deliver eTMF solutions that not only meet current demands but also anticipate future challenges in the dynamic landscape of clinical research and development. The projected market size is expected to reach several billion dollars by the end of the forecast period, underscoring the immense value and critical importance of eTMF systems in the pharmaceutical and biotechnology sectors.
Leading Regions, Countries, or Segments in Electronic Trial Master File Systems
The Electronic Trial Master File (eTMF) systems market showcases distinct leadership across various geographical regions and application segments, driven by a confluence of factors. North America, particularly the United States, consistently emerges as a dominant force due to its mature pharmaceutical industry, robust regulatory framework (FDA), and high adoption rate of advanced clinical technologies. The region's significant investment in research and development, coupled with a large number of ongoing clinical trials, fuels the demand for sophisticated eTMF software. Europe, with countries like Germany, the UK, and Switzerland, follows closely, benefiting from established life sciences hubs and proactive regulatory bodies like the EMA. The increasing number of multi-national clinical trials conducted in these regions further bolsters the market.
Within the application segments, hospitals represent a substantial driver of eTMF adoption. Their role as primary sites for conducting clinical trials necessitates efficient document management to ensure compliance with protocols and regulatory requirements. The increasing complexity of hospital operations and the growing volume of patient data handled daily make eTMF systems indispensable for maintaining organized and accessible trial documentation. Clinics, especially specialized research-focused ones, also demonstrate a strong demand for eTMF solutions, albeit with potentially different scaling and integration needs compared to larger hospital systems. The ability of eTMF systems to centralize information and facilitate remote access is particularly valuable for clinics that may have multiple locations or limited on-site administrative resources. Ambulatory care centers are also increasingly adopting eTMF solutions, recognizing the benefits of digital record-keeping for patient care and research participation.
Regarding deployment types, cloud-based eTMF solutions are rapidly gaining prominence, eclipsing on-premise deployments. The cloud offers unparalleled scalability, flexibility, and cost-effectiveness, aligning perfectly with the dynamic nature of clinical trials. Key drivers for cloud adoption include reduced IT infrastructure costs, faster deployment times, seamless updates, and enhanced accessibility for global research teams. The ability to access eTMF data from anywhere, at any time, is a significant advantage for geographically dispersed clinical research. Regulatory bodies are also becoming more comfortable with cloud-based solutions, provided they meet stringent data security and privacy standards. The market's preference for cloud underscores the industry's embrace of digital transformation and its commitment to leveraging advanced technologies for improved clinical trial management. This shift is projected to continue, further solidifying the dominance of cloud in the eTMF market.
- Dominant Region: North America (USA) and Europe.
- Key Drivers in North America: Strong regulatory environment (FDA), high R&D investment, numerous clinical trials.
- Key Drivers in Europe: Established life sciences clusters, proactive regulatory bodies (EMA), multinational trials.
- Leading Application Segment: Hospitals, followed by Clinics and Ambulatory Care Centers.
- Hospital eTMF Drivers: Need for efficient document management, compliance, patient data handling.
- Clinic eTMF Drivers: Centralized information, remote access, scalability.
- Ambulatory Care Center eTMF Drivers: Digital record-keeping benefits, research participation.
- Dominant Deployment Type: Cloud-based eTMF solutions.
- Cloud Adoption Drivers: Scalability, flexibility, cost-effectiveness, accessibility, faster deployment.
- On-Premise Segment: Declining but still relevant for organizations with specific security or legacy system requirements.
Electronic Trial Master File Systems Product Innovations
Product innovations in Electronic Trial Master File (eTMF) systems are revolutionizing clinical trial management. Companies are intensely focused on developing solutions with enhanced automation, intuitive user interfaces, and seamless integration capabilities. Key advancements include AI-powered quality control features that proactively identify potential errors or omissions in eTMF documentation, significantly reducing manual review efforts. Furthermore, the development of interoperable eTMF platforms that can effortlessly communicate with other clinical trial systems like EDC, CTMS, and ePRO is a major focus, creating a unified data ecosystem. Performance metrics are continually being improved, with faster document upload speeds, robust search functionalities, and real-time reporting dashboards becoming standard. Unique selling propositions often revolve around comprehensive audit trails, advanced security protocols, and flexible configuration options to meet diverse study requirements. These technological advancements are crucial for streamlining processes, ensuring data integrity, and accelerating the overall clinical trial lifecycle.
Propelling Factors for Electronic Trial Master File Systems Growth
The Electronic Trial Master File (eTMF) systems market is propelled by several key factors. Foremost is the increasing stringency and complexity of global regulatory requirements, compelling organizations to adopt robust systems for compliance and audit readiness. Technological advancements, particularly in cloud computing and AI, are enabling more efficient and accessible eTMF solutions. The growing trend towards decentralized clinical trials (DCTs) and hybrid models also necessitates remote access and real-time collaboration features inherent in advanced eTMF platforms. Furthermore, the constant pressure to accelerate drug development timelines and reduce operational costs drives the adoption of digitized trial management solutions like eTMF. Economic factors, including increased investment in pharmaceutical R&D and the growing number of clinical trials globally, also contribute significantly to market expansion, with the market size projected to reach billions.
- Regulatory Compliance: Essential for meeting stringent global standards and facilitating audits.
- Technological Advancements: AI, cloud computing, and advanced analytics enhance efficiency and accessibility.
- Rise of Decentralized Clinical Trials (DCTs): Demand for remote access and collaboration features.
- Cost and Time Pressures: Drive for efficiency and accelerated drug development timelines.
- Increased R&D Investment: Growing number of clinical trials globally fuels demand.
Obstacles in the Electronic Trial Master File Systems Market
Despite robust growth, the Electronic Trial Master File (eTMF) systems market faces several obstacles. Regulatory challenges persist, with varying interpretations and evolving guidelines across different regions, requiring continuous adaptation of eTMF systems. High implementation costs for some advanced solutions can be a barrier for smaller organizations or those with limited budgets. Data security concerns and the need to comply with diverse data privacy regulations (e.g., GDPR, HIPAA) add complexity to system design and deployment. Resistance to change from traditional paper-based processes and a lack of digital literacy among some personnel can hinder adoption. Interoperability issues with legacy systems and other clinical trial software can also create integration challenges, impacting seamless data flow. The competitive landscape, though growing, presents challenges in differentiating offerings and acquiring market share, with estimated competitive pressures impacting market dynamics.
Future Opportunities in Electronic Trial Master File Systems
The Electronic Trial Master File (eTMF) systems market is ripe with future opportunities. The continued expansion of decentralized clinical trials (DCTs) will drive demand for highly integrated and remote-accessible eTMF solutions. The integration of artificial intelligence and machine learning for automated data review, anomaly detection, and predictive analytics presents a significant growth avenue. Emerging markets in Asia-Pacific and Latin America, with their rapidly expanding pharmaceutical sectors, offer substantial untapped potential. Furthermore, the development of specialized eTMF solutions tailored for specific therapeutic areas or trial complexities will cater to niche market demands. The increasing focus on real-world evidence (RWE) and the need to manage associated data will also create new opportunities for eTMF platforms to evolve and incorporate RWE management capabilities, leading to multi-billion dollar revenue streams.
Major Players in the Electronic Trial Master File Systems Ecosystem
Aurea Software Oracle Ennov Wingspan Covance Inc TransPerfect Phlexglobal SureClinical Inc Veeva Systems ePharmaSolutions MasterControl, Inc
Key Developments in Electronic Trial Master File Systems Industry
- January 2024: Veeva Systems launched a new module for AI-powered eTMF review, enhancing automated quality control.
- November 2023: Oracle announced significant upgrades to its eTMF solution, focusing on improved interoperability with its cloud-based clinical platform.
- July 2023: Ennov released an updated version of its eTMF system with advanced features for decentralized clinical trials.
- April 2023: TransPerfect acquired a key eTMF technology provider, expanding its service offerings in clinical trial documentation.
- February 2023: MasterControl, Inc. partnered with a major CRO to implement its eTMF solution across a portfolio of global studies.
- October 2022: Phlexglobal introduced enhanced analytics capabilities within its eTMF platform for better trial oversight.
- June 2022: SureClinical Inc. received significant investment to scale its cloud-based eTMF offerings.
- January 2022: ePharmaSolutions expanded its service model to include comprehensive eTMF management for smaller biotech companies.
Strategic Electronic Trial Master File Systems Market Forecast
The Electronic Trial Master File (eTMF) systems market is poised for substantial growth in the coming years, driven by strategic initiatives and evolving industry needs. The forecast indicates continued strong adoption of cloud-based eTMF solutions due to their inherent flexibility and scalability, catering to the increasing complexity of clinical trials. Investments in AI and machine learning for automated data validation and quality checks will become more prevalent, significantly boosting efficiency. The expansion of decentralized and hybrid clinical trial models will further propel demand for eTMF systems that offer seamless remote access and collaboration capabilities. Emerging markets present considerable untapped potential, offering new avenues for growth and market penetration. The overall market trajectory points towards continued innovation and increasing value, with the market size expected to reach several billion dollars.
Electronic Trial Master File Systems Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Ambulatory Care Centers
-
2. Types
- 2.1. On Premise
- 2.2. Cloud
Electronic Trial Master File Systems Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
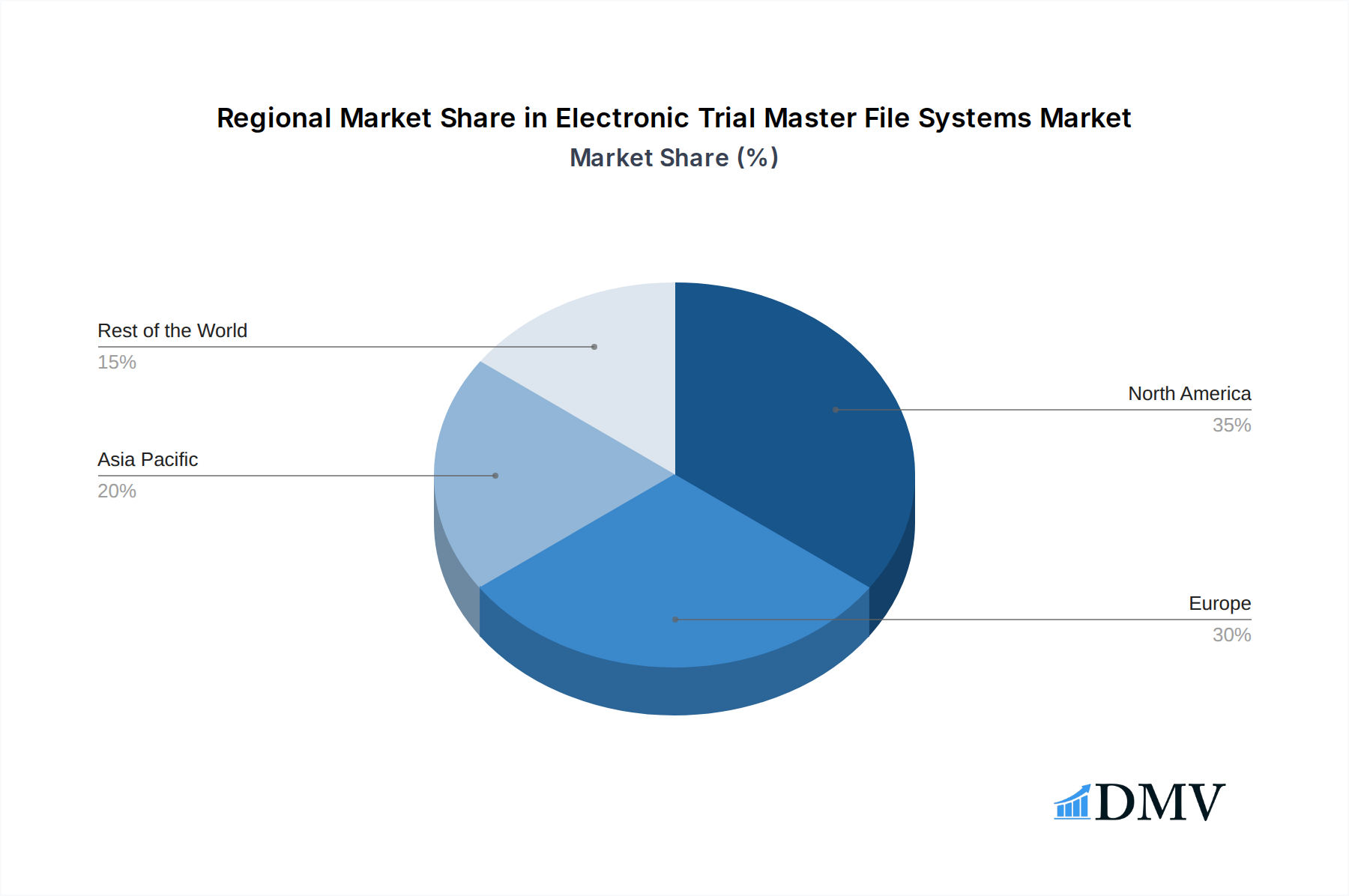
Electronic Trial Master File Systems Regional Market Share

Geographic Coverage of Electronic Trial Master File Systems
Electronic Trial Master File Systems REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Electronic Trial Master File Systems Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Ambulatory Care Centers
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. On Premise
- 5.2.2. Cloud
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Electronic Trial Master File Systems Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.1.3. Ambulatory Care Centers
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. On Premise
- 6.2.2. Cloud
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Electronic Trial Master File Systems Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.1.3. Ambulatory Care Centers
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. On Premise
- 7.2.2. Cloud
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Electronic Trial Master File Systems Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.1.3. Ambulatory Care Centers
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. On Premise
- 8.2.2. Cloud
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Electronic Trial Master File Systems Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.1.3. Ambulatory Care Centers
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. On Premise
- 9.2.2. Cloud
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Electronic Trial Master File Systems Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.1.3. Ambulatory Care Centers
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. On Premise
- 10.2.2. Cloud
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Aurea Software
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Oracle
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Ennov
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Wingspan
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Covance Inc
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 TransPerfect
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Phlexglobal
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 SureClinical Inc
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Veeva Systems
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 ePharmaSolutions
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 MasterControl
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Inc
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.1 Aurea Software
List of Figures
- Figure 1: Global Electronic Trial Master File Systems Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Electronic Trial Master File Systems Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Electronic Trial Master File Systems Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Electronic Trial Master File Systems Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Electronic Trial Master File Systems Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Electronic Trial Master File Systems Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Electronic Trial Master File Systems Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Electronic Trial Master File Systems Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Electronic Trial Master File Systems Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Electronic Trial Master File Systems Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Electronic Trial Master File Systems Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Electronic Trial Master File Systems Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Electronic Trial Master File Systems Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Electronic Trial Master File Systems Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Electronic Trial Master File Systems Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Electronic Trial Master File Systems Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Electronic Trial Master File Systems Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Electronic Trial Master File Systems Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Electronic Trial Master File Systems Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Electronic Trial Master File Systems Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Electronic Trial Master File Systems Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Electronic Trial Master File Systems Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Electronic Trial Master File Systems Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Electronic Trial Master File Systems Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Electronic Trial Master File Systems Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Electronic Trial Master File Systems Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Electronic Trial Master File Systems Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Electronic Trial Master File Systems Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Electronic Trial Master File Systems Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Electronic Trial Master File Systems Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Electronic Trial Master File Systems Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Electronic Trial Master File Systems Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Electronic Trial Master File Systems Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Electronic Trial Master File Systems?
The projected CAGR is approximately 12.8%.
2. Which companies are prominent players in the Electronic Trial Master File Systems?
Key companies in the market include Aurea Software, Oracle, Ennov, Wingspan, Covance Inc, TransPerfect, Phlexglobal, SureClinical Inc, Veeva Systems, ePharmaSolutions, MasterControl, Inc.
3. What are the main segments of the Electronic Trial Master File Systems?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Electronic Trial Master File Systems," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Electronic Trial Master File Systems report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Electronic Trial Master File Systems?
To stay informed about further developments, trends, and reports in the Electronic Trial Master File Systems, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


