Key Insights
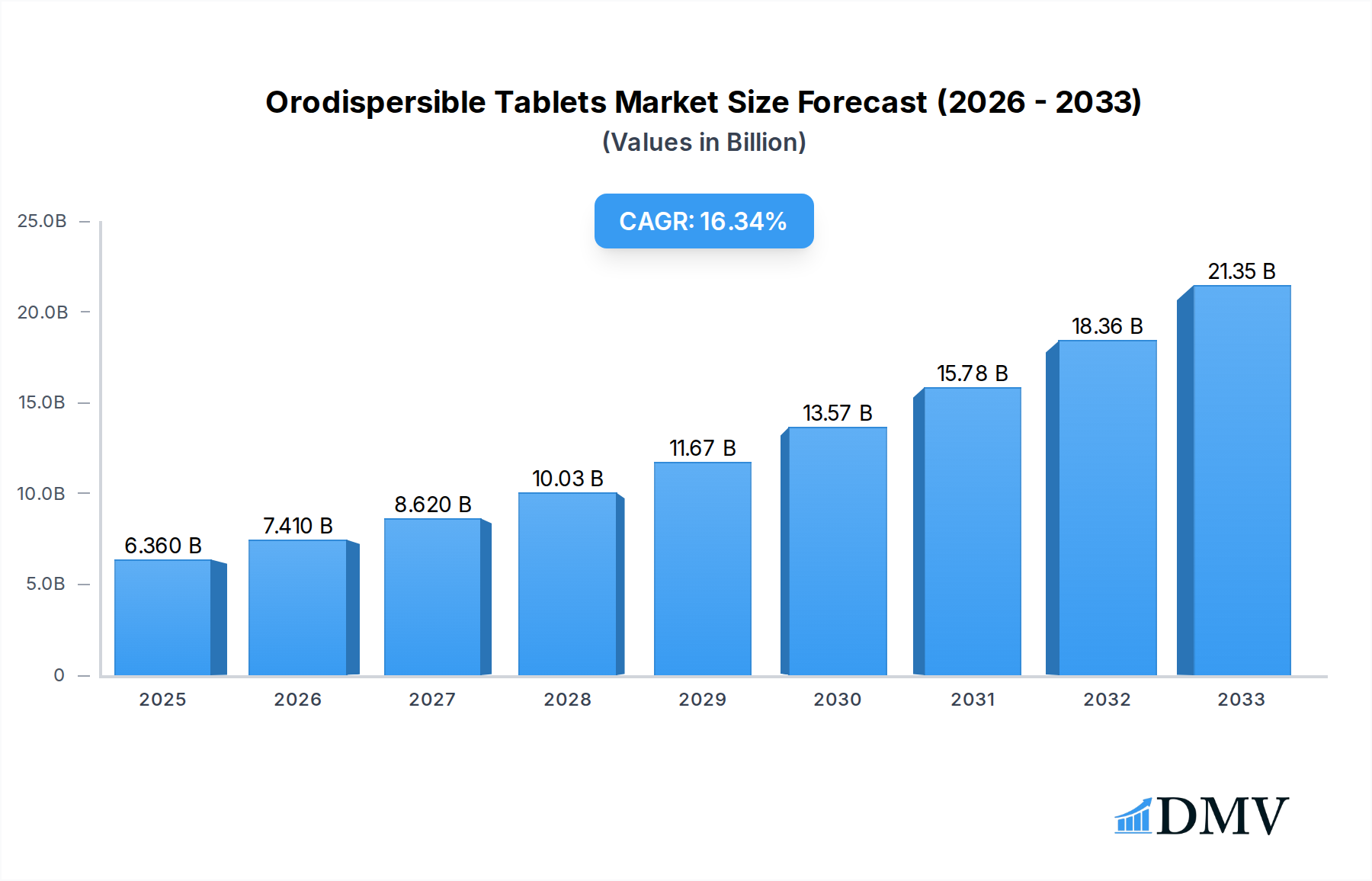
The global Orodispersible Tablets market is poised for significant expansion, projected to reach USD 6.36 billion in 2025, demonstrating a robust CAGR of 16.25% through the forecast period. This rapid growth is primarily fueled by the increasing prevalence of chronic diseases, particularly CNS and gastrointestinal disorders, which necessitate convenient and faster-acting therapeutic solutions. Orodispersible tablets, also known as orally disintegrating tablets (ODTs), offer distinct advantages over conventional oral dosage forms, including rapid onset of action, improved patient compliance, and suitability for individuals with swallowing difficulties. The expanding elderly population, coupled with a growing awareness of the benefits of ODTs, further propels market demand. Key drivers include advancements in pharmaceutical formulation technologies, the development of novel drug delivery systems, and the increasing adoption of ODTs for various therapeutic applications, from antipsychotics and anti-epileptics to treatments for cardiovascular and gastrointestinal ailments. The market is witnessing a surge in research and development efforts aimed at expanding the therapeutic scope of ODTs and enhancing their stability and efficacy.
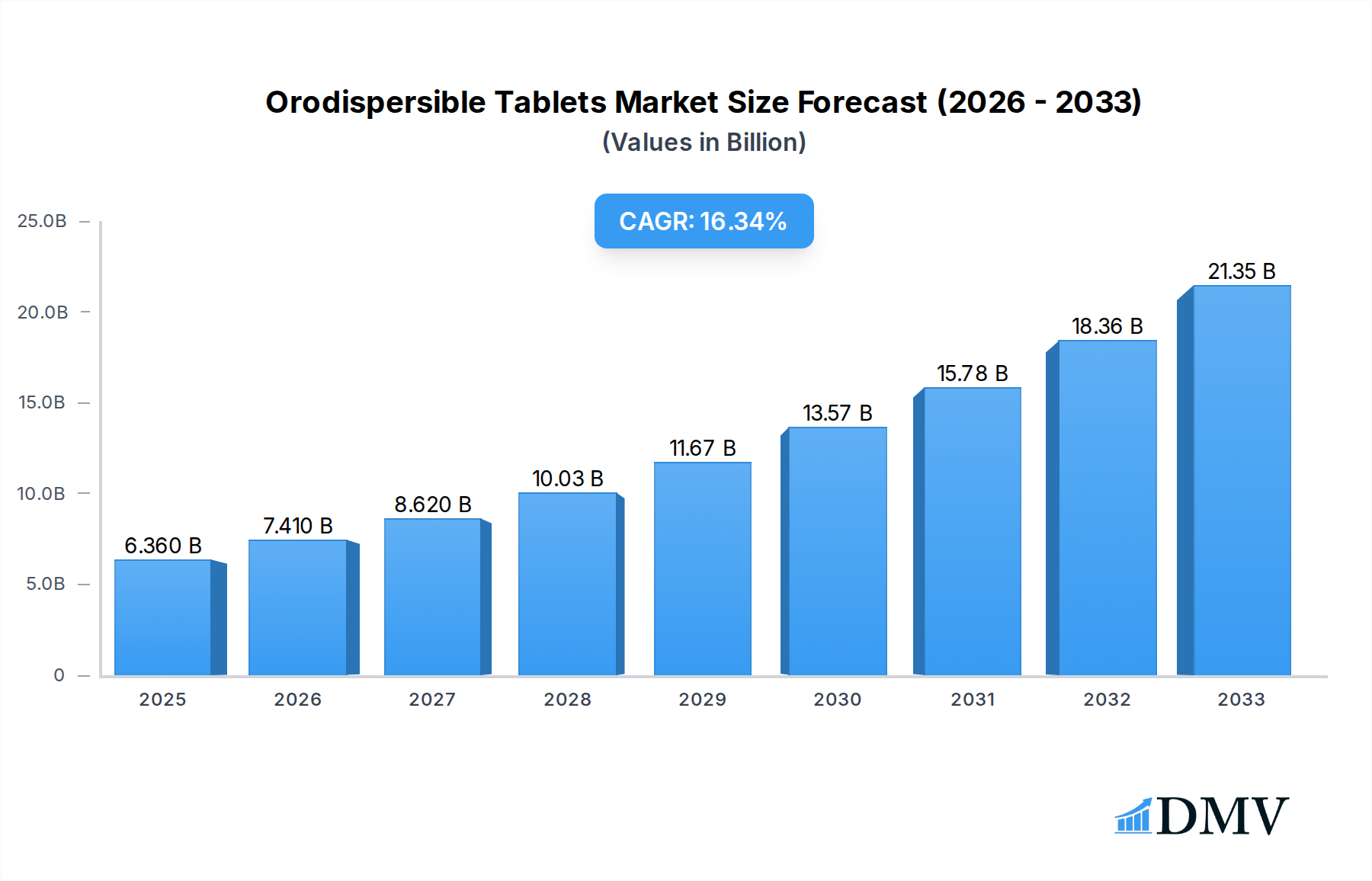
Orodispersible Tablets Market Size (In Billion)

The market segmentation reveals a dynamic landscape, with CNS Diseases and Gastrointestinal Diseases emerging as prominent application segments due to their high patient burden and the inherent advantages of ODTs in managing these conditions. In terms of types, Anti-Psychotics Drugs and Anti-Epileptics Drugs are expected to witness substantial growth, driven by the critical need for improved treatment adherence in patients with these neurological and psychiatric disorders. Major pharmaceutical players like Teva, Merck, Pfizer, and Johnson & Johnson are actively investing in R&D and strategic collaborations to capitalize on this burgeoning market. Geographically, North America and Europe are leading the market, owing to their well-established healthcare infrastructure, high healthcare spending, and early adoption of advanced pharmaceutical formulations. However, the Asia Pacific region is anticipated to exhibit the fastest growth, driven by its large patient population, increasing disposable incomes, and a growing focus on improving healthcare accessibility and quality. The market's trajectory is further influenced by supportive regulatory frameworks and the increasing preference of healthcare professionals and patients for ODTs.
Orodispersible Tablets Company Market Share

This in-depth market research report provides a definitive analysis of the global Orodispersible Tablets market, offering a comprehensive overview of its current landscape, historical performance, and projected trajectory. With a study period spanning from 2019 to 2033, a base year of 2025, and a forecast period from 2025 to 2033, this report leverages extensive data and expert insights to empower stakeholders with actionable intelligence. Our analysis delves into market concentration, key industry developments, competitive dynamics, and the evolving needs of patients and healthcare providers. The report is meticulously crafted to address the critical questions surrounding this rapidly growing pharmaceutical segment, particularly for oral disintegrating tablets, fast-dissolving tablets, and orally disintegrating formulations.
Orodispersible Tablets Market Composition & Trends
The global Orodispersible Tablets market exhibits a dynamic and moderately concentrated composition, driven by continuous innovation and strategic collaborations among major pharmaceutical players. Innovation catalysts such as advancements in drug delivery technologies, patient convenience, and improved therapeutic efficacy are profoundly shaping market trends. The regulatory landscape, while stringent, is progressively adapting to facilitate the adoption of novel ODT formulations. Substitute products, primarily conventional tablets and capsules, are facing increasing competition from ODTs due to their superior patient compliance, especially for pediatric and geriatric populations, and for conditions requiring rapid onset of action.
End-user profiles are diverse, encompassing patients with:
- CNS Diseases: Requiring quick relief from neurological symptoms.
- Gastrointestinal Diseases: Benefiting from faster absorption and reduced gastrointestinal irritation.
- CVS Diseases: Where rapid drug delivery is crucial for managing critical conditions.
- Other Applications: Including pain management, allergies, and motion sickness.
Mergers and Acquisitions (M&A) activities are a significant indicator of market maturity and strategic positioning. The total value of M&A deals in the historical period (2019-2024) is estimated to be in the billions, with key players actively consolidating their market presence and expanding their product portfolios. For instance, strategic acquisitions aimed at securing patented ODT technologies or expanding into niche therapeutic areas have been prevalent. Market share distribution, while varying by segment and region, indicates a healthy competition, with specialized ODT manufacturers and large pharmaceutical conglomerates vying for dominance. The market is projected to witness a significant CAGR, exceeding XX% in the forecast period, driven by increasing demand for patient-centric drug delivery systems.
Orodispersible Tablets Industry Evolution
The Orodispersible Tablets industry has undergone a remarkable evolution, transforming from a niche segment to a mainstream pharmaceutical solution. This evolution is characterized by persistent market growth trajectories fueled by unmet patient needs and technological breakthroughs. Historically, from 2019 to 2024, the market experienced steady expansion, with a compound annual growth rate (CAGR) of approximately XX%. The base year of 2025 positions the market at an estimated value of billions, with robust projections for the forecast period (2025-2033) anticipating a CAGR of over XX%, reaching trillions by 2033.
Technological advancements have been pivotal in this evolution. Early advancements focused on overcoming the challenges of rapid disintegration and drug palatability. More recent innovations have centered on enhancing bioavailability, developing ODTs for complex molecules, and improving manufacturing processes for cost-effectiveness and scalability. The adoption of advanced excipients, such as superdisintegrants and taste-masking agents, has significantly improved the quality and patient acceptance of these formulations.
Shifting consumer demands, particularly the growing preference for patient-friendly and convenient dosage forms, have been a major impetus. Patients are increasingly seeking alternatives to traditional pills that require water or present swallowing difficulties. This demand is amplified by an aging global population and a rising prevalence of chronic diseases. The focus on improving patient compliance and adherence to treatment regimens has positioned ODTs as a preferred choice across a spectrum of therapeutic areas, including CNS diseases, gastrointestinal disorders, and cardiovascular conditions.
The industry's journey reflects a strategic shift towards patient-centric drug delivery, leading to substantial investments in research and development. This has resulted in the introduction of a wider range of therapeutic agents in ODT formats, from anti-psychotics and anti-epileptics to pain relievers and anti-allergy medications. The market's evolution is a testament to the successful integration of pharmaceutical science, patient advocacy, and market demand, paving the way for continued growth and innovation.
Leading Regions, Countries, or Segments in Orodispersible Tablets
The Orodispersible Tablets market exhibits a pronounced leadership across specific regions and therapeutic segments, driven by a confluence of factors including healthcare infrastructure, disease prevalence, and regulatory support. North America, particularly the United States, has consistently emerged as a dominant force due to its advanced healthcare system, high patient awareness, and substantial investment in pharmaceutical R&D. The presence of major pharmaceutical companies like Pfizer, Johnson & Johnson, and Eli Lilly and Company, actively involved in the development and commercialization of ODTs, further solidifies its leading position.
Within the Application segment, CNS Diseases represent a critical area of dominance for Orodispersible Tablets. The need for rapid and reliable drug delivery to manage neurological conditions such as epilepsy, Parkinson's disease, and psychiatric disorders necessitates fast-acting formulations. ODTs offer significant advantages in terms of faster absorption and bypassing the initial stages of digestion, making them ideal for patients who may have difficulty swallowing or require immediate symptom relief.
In terms of Types, Anti-Epileptics Drugs and Anti-Psychotics Drugs are key segments driving ODT adoption within CNS diseases. For instance, the development of orally disintegrating anti-epileptic drugs has greatly improved seizure management and patient adherence, particularly in emergency situations. Similarly, orally disintegrating antipsychotic medications have been instrumental in enhancing treatment compliance and reducing the stigma associated with certain psychiatric conditions.
Key drivers contributing to this dominance include:
- High Prevalence of CNS Disorders: North America and Europe have a significant burden of neurological and psychiatric diseases, creating a substantial demand for effective treatment options.
- Technological Advancements in Drug Delivery: Companies are heavily investing in novel ODT technologies to enhance bioavailability, palatability, and disintegration times for a wider range of APIs.
- Favorable Regulatory Landscape: Regulatory bodies in leading regions often encourage the development of patient-friendly dosage forms, streamlining approval processes for innovative ODTs.
- Patient Centricity: A growing emphasis on patient convenience and adherence is a major driver, with ODTs offering a clear advantage over traditional oral dosage forms for many patients.
- Strategic Collaborations and Investments: Pharmaceutical giants and specialized ODT manufacturers are actively collaborating and investing billions in R&D and market expansion.
- Market Size and Healthcare Spending: Higher healthcare expenditure and robust insurance coverage in developed nations enable greater access and demand for advanced pharmaceutical products like ODTs.
The market for CNS disease-related ODTs is projected to continue its upward trajectory, supported by ongoing research into new drug formulations and expanding indications. The estimated market size for ODTs in CNS applications alone is expected to reach billions by 2025, with strong growth anticipated through 2033.
Orodispersible Tablets Product Innovations
Product innovations in Orodispersible Tablets are revolutionizing drug delivery, focusing on enhanced patient experience and therapeutic efficacy. Unique selling propositions include rapid disintegration within seconds of contact with saliva, eliminating the need for water and facilitating administration in diverse settings. Technological advancements are centered on improving taste masking of bitter APIs, increasing drug loading capacity, and developing ODTs for challenging molecules. For example, advancements in spray-drying and lyophilization techniques have enabled the production of ODTs with improved dissolution profiles and greater stability, making them suitable for sensitive compounds. These innovations translate into improved patient compliance, particularly for pediatric, geriatric, and dysphagic populations, thereby enhancing treatment outcomes and contributing to a projected market value in the billions.
Propelling Factors for Orodispersible Tablets Growth
The growth of the Orodispersible Tablets market is propelled by several key factors. Firstly, technological advancements in drug delivery systems, particularly in formulation science and manufacturing processes, have made ODTs more viable and cost-effective. Secondly, growing patient demand for convenience and adherence is a significant driver, as ODTs eliminate the need for water and are easier to swallow, especially for pediatric and geriatric populations. Thirdly, favorable regulatory support in various regions, encouraging the development of patient-centric drug delivery solutions, further bolsters market expansion. The increasing prevalence of chronic diseases and the need for rapid onset of action in certain therapeutic areas, such as CNS disorders, also contribute substantially to the market's upward trajectory, with the market expected to reach billions in the coming years.
Obstacles in the Orodispersible Tablets Market
Despite robust growth, the Orodispersible Tablets market faces several obstacles. Manufacturing complexities and higher production costs compared to conventional tablets can limit scalability and affordability. Regulatory hurdles related to taste masking, stability, and bioavailability can prolong the development timeline and increase R&D expenses, impacting the market's accessibility. Supply chain disruptions, as witnessed in recent global events, can affect the availability of crucial excipients and raw materials, potentially leading to delays and increased costs. Furthermore, intense competition from established conventional dosage forms and the need for significant patient and physician education on the benefits of ODTs present ongoing challenges, with potential impacts on market penetration estimated in the billions of dollars for missed opportunities.
Future Opportunities in Orodispersible Tablets
The future of the Orodispersible Tablets market is brimming with opportunities. Emerging opportunities lie in the development of ODTs for complex biologics and peptide-based drugs, expanding the therapeutic reach beyond small molecules. The growing demand for personalized medicine will drive the development of ODTs for targeted patient groups and specific disease indications. Furthermore, the expansion into emerging markets, where access to clean water is limited or patient convenience is highly valued, presents a significant growth avenue, potentially adding billions to the market's global footprint. Innovations in drug-in-device technologies and advancements in taste-masking techniques will further enhance patient acceptance and broaden the application spectrum, opening up new revenue streams in the billions.
Major Players in the Orodispersible Tablets Ecosystem
- Teva
- Merck
- Mylan
- Pfizer
- Johnson & Johnson
- GSK
- Otsuka
- Eli Lilly and Company
- AstraZeneca
- Bristol-Myers Squibb
- Conquer
Key Developments in Orodispersible Tablets Industry
- 2023 Q4: Launch of a new orally disintegrating anti-epileptic drug formulation by Merck, targeting improved seizure control and patient compliance, contributing billions to the company's revenue.
- 2023 Q3: Teva Pharmaceutical Industries announced strategic partnerships for the development of novel ODTs for CNS disorders, investing billions in R&D.
- 2023 Q2: Mylan (now part of Viatris) expanded its portfolio of generic orally disintegrating tablets, focusing on cardiovascular disease treatments, reinforcing its market share in the billions.
- 2023 Q1: Pfizer reported significant clinical trial success for an orally disintegrating antipsychotic drug, anticipating substantial market uptake and billions in future sales.
- 2022 Q4: Johnson & Johnson received regulatory approval for an orally disintegrating pain management medication, marking a significant advancement in patient-friendly pain relief solutions, estimated to impact the market by billions.
- 2022 Q3: GSK unveiled innovative ODT technology aimed at improving the palatability and bioavailability of a wide range of APIs, with projections of adding billions to its pharmaceutical segment.
- 2022 Q2: Otsuka Pharmaceutical received expanded indications for an existing orally disintegrating drug, further solidifying its presence in the psychiatric disorders market, contributing billions to its financial performance.
- 2022 Q1: Eli Lilly and Company announced substantial investments in ODT manufacturing capabilities, anticipating increased demand and a market expansion worth billions.
- 2021 Q4: AstraZeneca explored novel drug delivery systems for oncology, including potential ODT applications, indicating a strategic interest in this billion-dollar market segment.
- 2021 Q3: Bristol-Myers Squibb highlighted the growing importance of ODTs in patient adherence for chronic disease management, with a focus on cardiovascular therapies, impacting billions in market value.
Strategic Orodispersible Tablets Market Forecast
The strategic Orodispersible Tablets market forecast indicates a robust expansion, driven by continuous innovation and an increasing emphasis on patient-centric healthcare solutions. The projected growth is underpinned by the sustained demand for fast-dissolving tablets and oral disintegrating formulations across a multitude of therapeutic applications, including CNS diseases, gastrointestinal disorders, and cardiovascular conditions. Investments in advanced manufacturing technologies and the development of ODTs for challenging APIs will further enhance market penetration. The strategic initiatives by major players like Pfizer, Teva, and Johnson & Johnson, coupled with favorable regulatory environments, are expected to propel the market value into the trillions by the end of the forecast period, offering significant opportunities for stakeholders across the pharmaceutical value chain.
Orodispersible Tablets Segmentation
-
1. Application
- 1.1. CNS Diseases
- 1.2. Gastrointestinal Diseases
- 1.3. CVS Diseases
- 1.4. Other
-
2. Types
- 2.1. Anti-Psychotics Drug
- 2.2. Anti-Epileptics Drug
- 2.3. Other
Orodispersible Tablets Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
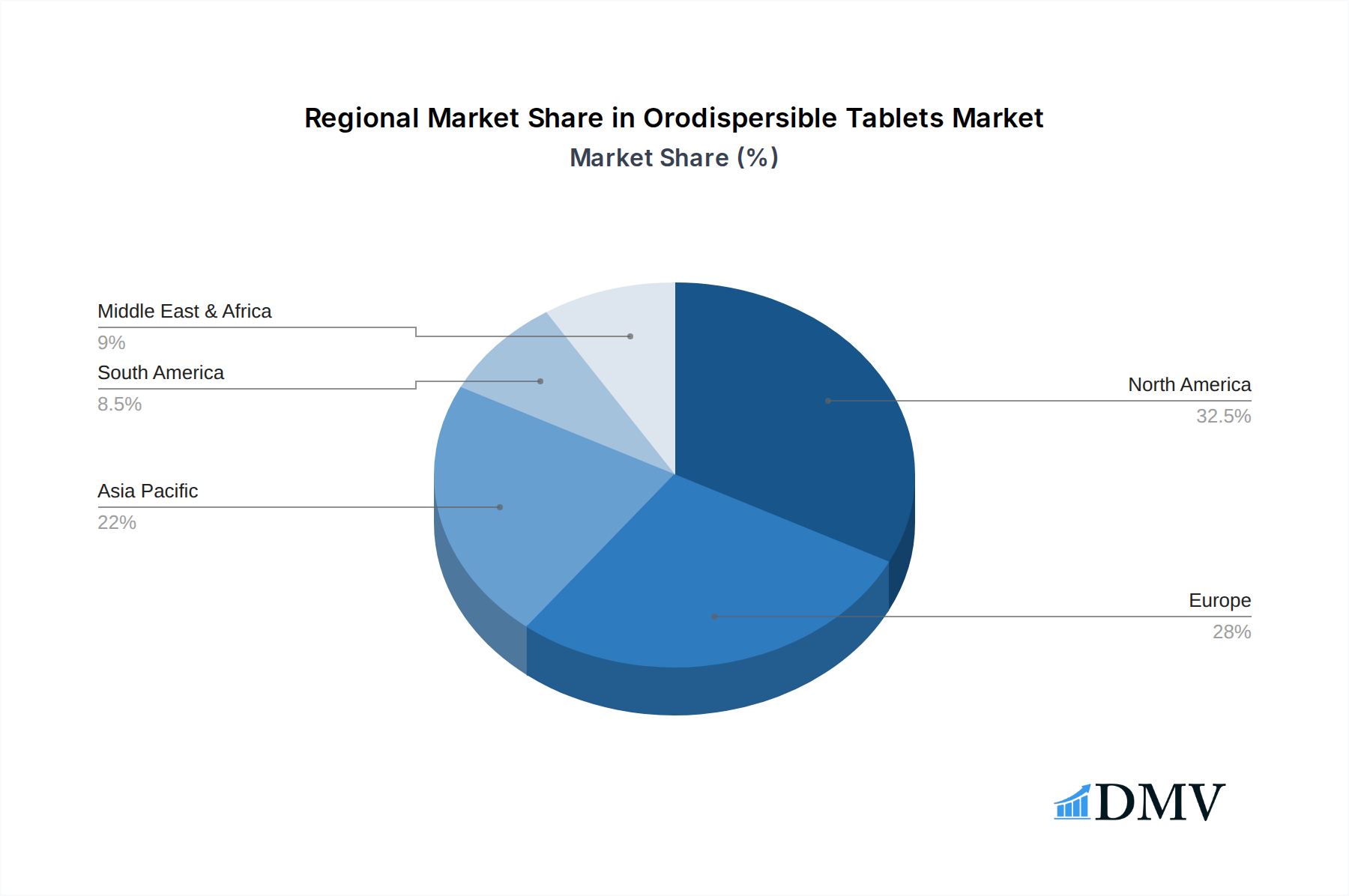
Orodispersible Tablets Regional Market Share

Geographic Coverage of Orodispersible Tablets
Orodispersible Tablets REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 16.25% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Orodispersible Tablets Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. CNS Diseases
- 5.1.2. Gastrointestinal Diseases
- 5.1.3. CVS Diseases
- 5.1.4. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Anti-Psychotics Drug
- 5.2.2. Anti-Epileptics Drug
- 5.2.3. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Orodispersible Tablets Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. CNS Diseases
- 6.1.2. Gastrointestinal Diseases
- 6.1.3. CVS Diseases
- 6.1.4. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Anti-Psychotics Drug
- 6.2.2. Anti-Epileptics Drug
- 6.2.3. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Orodispersible Tablets Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. CNS Diseases
- 7.1.2. Gastrointestinal Diseases
- 7.1.3. CVS Diseases
- 7.1.4. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Anti-Psychotics Drug
- 7.2.2. Anti-Epileptics Drug
- 7.2.3. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Orodispersible Tablets Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. CNS Diseases
- 8.1.2. Gastrointestinal Diseases
- 8.1.3. CVS Diseases
- 8.1.4. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Anti-Psychotics Drug
- 8.2.2. Anti-Epileptics Drug
- 8.2.3. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Orodispersible Tablets Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. CNS Diseases
- 9.1.2. Gastrointestinal Diseases
- 9.1.3. CVS Diseases
- 9.1.4. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Anti-Psychotics Drug
- 9.2.2. Anti-Epileptics Drug
- 9.2.3. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Orodispersible Tablets Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. CNS Diseases
- 10.1.2. Gastrointestinal Diseases
- 10.1.3. CVS Diseases
- 10.1.4. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Anti-Psychotics Drug
- 10.2.2. Anti-Epileptics Drug
- 10.2.3. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Teva
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Merck
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Mylan
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Pfizer
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Johnson and Johnson
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 GSK
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Otsuka
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Eli Lilly and Company
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 AstraZeneca
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Bristol-Myers Squibb
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Conquer
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.1 Teva
List of Figures
- Figure 1: Global Orodispersible Tablets Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Orodispersible Tablets Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Orodispersible Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Orodispersible Tablets Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Orodispersible Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Orodispersible Tablets Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Orodispersible Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Orodispersible Tablets Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Orodispersible Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Orodispersible Tablets Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Orodispersible Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Orodispersible Tablets Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Orodispersible Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Orodispersible Tablets Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Orodispersible Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Orodispersible Tablets Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Orodispersible Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Orodispersible Tablets Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Orodispersible Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Orodispersible Tablets Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Orodispersible Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Orodispersible Tablets Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Orodispersible Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Orodispersible Tablets Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Orodispersible Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Orodispersible Tablets Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Orodispersible Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Orodispersible Tablets Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Orodispersible Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Orodispersible Tablets Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Orodispersible Tablets Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Orodispersible Tablets Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Orodispersible Tablets Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Orodispersible Tablets Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Orodispersible Tablets Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Orodispersible Tablets Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Orodispersible Tablets Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Orodispersible Tablets Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Orodispersible Tablets Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Orodispersible Tablets Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Orodispersible Tablets Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Orodispersible Tablets Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Orodispersible Tablets Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Orodispersible Tablets Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Orodispersible Tablets Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Orodispersible Tablets Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Orodispersible Tablets Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Orodispersible Tablets Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Orodispersible Tablets Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Orodispersible Tablets Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Orodispersible Tablets?
The projected CAGR is approximately 16.25%.
2. Which companies are prominent players in the Orodispersible Tablets?
Key companies in the market include Teva, Merck, Mylan, Pfizer, Johnson and Johnson, GSK, Otsuka, Eli Lilly and Company, AstraZeneca, Bristol-Myers Squibb, Conquer.
3. What are the main segments of the Orodispersible Tablets?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 6.36 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Orodispersible Tablets," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Orodispersible Tablets report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Orodispersible Tablets?
To stay informed about further developments, trends, and reports in the Orodispersible Tablets, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


