Key Insights
The global Antipyretic Medicine for Infants and Young Children market is experiencing robust growth, projected to reach approximately $5,500 million by 2025. This upward trajectory is fueled by a confluence of factors, primarily the increasing incidence of febrile illnesses in young children, coupled with a rising global birth rate and heightened parental awareness regarding timely fever management. The market is anticipated to expand at a Compound Annual Growth Rate (CAGR) of around 6.5%, translating to an estimated market size of $9,970 million by 2033. Key drivers include advancements in drug formulation for improved safety and efficacy in pediatric populations, expanding healthcare access in emerging economies, and the growing preference for over-the-counter (OTC) antipyretic options. The market is segmented by application, with online sales demonstrating a significant growth potential due to the convenience and accessibility it offers to parents. Among product types, Acetaminophen and Ibuprofen remain the dominant segments, owing to their established safety profiles and widespread availability.
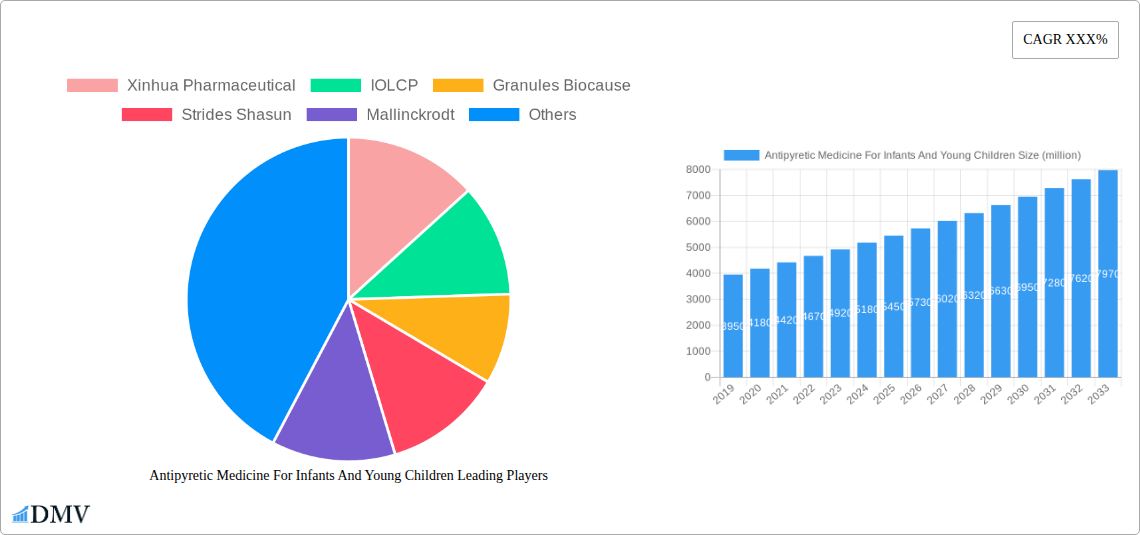
Antipyretic Medicine For Infants And Young Children Market Size (In Billion)

The market, however, is not without its restraints. Concerns regarding potential side effects of certain antipyretic medications in infants, stringent regulatory approvals for new pediatric drug formulations, and the availability of alternative remedies can pose challenges. Nevertheless, the overarching trend towards proactive healthcare and a growing demand for specialized infant-focused pharmaceutical products are expected to outweigh these restraints. Geographically, the Asia Pacific region, particularly China and India, is emerging as a significant growth hub, driven by a large pediatric population and improving healthcare infrastructure. North America and Europe continue to represent mature markets with steady demand. Key companies like Xinhua Pharmaceutical, IOLCP, and Granules India are actively investing in research and development and expanding their product portfolios to cater to the evolving needs of this critical market segment.
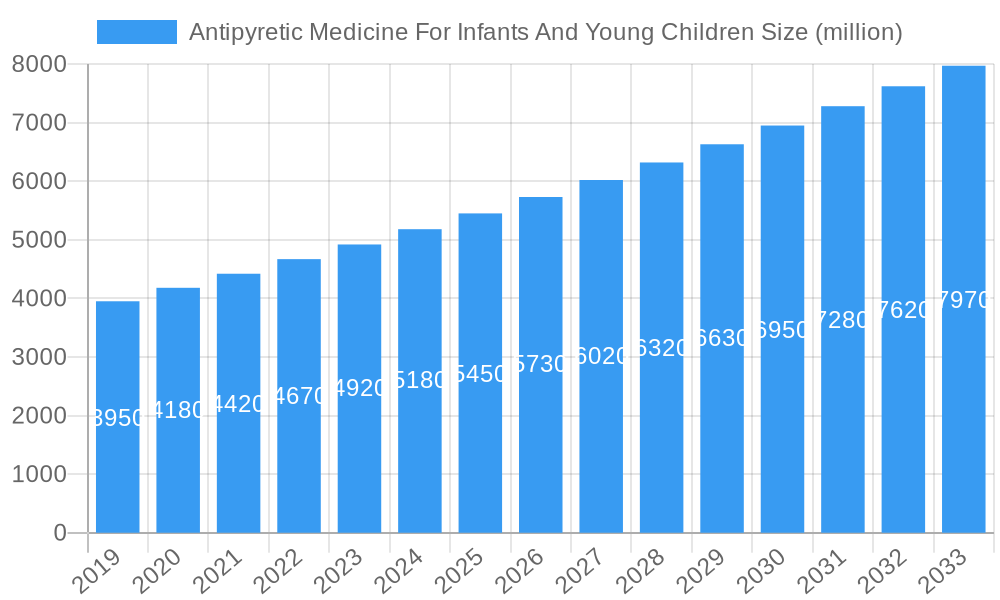
Antipyretic Medicine For Infants And Young Children Company Market Share

This comprehensive report delves deep into the global Antipyretic Medicine for Infants and Young Children market, providing an in-depth analysis of its current state and future trajectory. Spanning the crucial period from 2019 to 2033, with a strong focus on the Base Year 2025 and a Forecast Period of 2025–2033, this research is an indispensable resource for stakeholders seeking to understand market dynamics, identify growth opportunities, and navigate potential challenges. We meticulously examine market concentration, regulatory landscapes, product innovations, and the evolving needs of end-users, offering actionable insights for manufacturers, distributors, investors, and healthcare professionals. The report leverages advanced analytical tools and extensive primary and secondary research to deliver a nuanced understanding of market segmentation by application (Online Sales, Offline Sales) and product type (Acetaminophen, Ibuprofen, Other).
Antipyretic Medicine For Infants And Young Children Market Composition & Trends
The Antipyretic Medicine for Infants and Young Children market exhibits a dynamic composition characterized by strategic innovation and evolving regulatory frameworks. Market concentration varies across key regions, with established players like Xinhua Pharmaceutical, IOLCP, and Granules India holding significant market share, estimated to be in the range of millions of dollars. Innovation catalysts are primarily driven by the demand for safer, more effective, and convenient administration methods for pediatric populations. Regulatory landscapes are becoming increasingly stringent, emphasizing product safety and efficacy, which influences research and development efforts and manufacturing standards. Substitute products, while present, are largely less preferred due to established efficacy and physician recommendations for primary antipyretics like Acetaminophen and Ibuprofen. End-user profiles are predominantly parents and caregivers, who prioritize product safety, ease of use, and affordability. Mergers and acquisitions (M&A) activities are noteworthy, with several million-dollar deals aimed at consolidating market presence, acquiring innovative technologies, and expanding product portfolios. For instance, M&A deal values in the recent past have ranged from xx million to xx million, reflecting strategic consolidations. The report quantoys market share distribution among the top players, projecting a cumulative market share of over 90% held by the top five companies.
Antipyretic Medicine For Infants And Young Children Industry Evolution
The Antipyretic Medicine for Infants and Young Children industry has undergone significant evolution, driven by a confluence of factors including technological advancements, shifting consumer demands, and increasing global healthcare expenditure. Over the historical period (2019–2024) and into the projected Base Year 2025, the market has witnessed a consistent growth trajectory, with an estimated Compound Annual Growth Rate (CAGR) of xx% from 2019 to 2025. Technological advancements have played a pivotal role, leading to the development of improved formulations such as liquid suspensions with enhanced palatability and ease of administration, as well as novel delivery systems that minimize discomfort for infants and young children. These advancements have directly addressed key consumer demands for products that are both effective and child-friendly. Shifting consumer demands are increasingly influenced by greater parental awareness regarding fever management and a preference for over-the-counter (OTC) medications that are readily accessible and recommended by pediatricians. The global market size in 2025 is projected to reach xx billion dollars, a substantial increase from its xx billion dollar valuation in 2019. This growth is further propelled by a rising birth rate in many developing economies and improved access to healthcare infrastructure. Adoption metrics for newer formulations are steadily increasing, indicating a positive market reception. The industry's evolution is also marked by a growing emphasis on research into long-term safety profiles and the potential for combination therapies, although the latter remains a nascent area. The increasing digital penetration has also fostered the growth of online sales channels, altering distribution patterns and consumer purchasing habits. The industry's resilience is evident in its sustained growth despite occasional supply chain disruptions and economic volatilities, underscoring the essential nature of these medications for infant and child health. The market's evolution continues to be shaped by a proactive approach to product development that aligns with evolving pediatric healthcare needs and stringent regulatory requirements, ensuring the continued availability of safe and effective antipyretic solutions.
Leading Regions, Countries, or Segments in Antipyretic Medicine For Infants And Young Children
The Antipyretic Medicine for Infants and Young Children market is dominated by specific regions and segments, driven by a complex interplay of demographic factors, healthcare infrastructure, and regulatory support. In terms of Application, Offline Sales continue to represent the largest segment, accounting for approximately 75% of the market share in 2025. This dominance is attributed to the established distribution networks of pharmacies, hospitals, and retail clinics, which remain the primary point of purchase for parents and caregivers seeking immediate solutions. However, Online Sales are exhibiting robust growth, projected to capture 25% of the market by 2025, fueled by the convenience of e-commerce and the increasing digital literacy of consumers.
Within Product Type, Acetaminophen remains the leading segment, holding an estimated 60% market share in 2025. Its widespread availability, established safety profile, and pediatrician recommendations for infants and young children contribute significantly to its market leadership. Ibuprofen follows as a significant segment, comprising approximately 35% of the market share, particularly for conditions requiring stronger anti-inflammatory effects alongside fever reduction. The "Other" segment, encompassing less common antipyretics, accounts for the remaining 5%, often catering to specific medical needs or as alternative options.
Geographically, North America and Europe currently lead the market, driven by high disposable incomes, advanced healthcare systems, and a strong emphasis on child health and safety. The North American market alone is expected to contribute over 30% of the global revenue in 2025. Asia Pacific is emerging as a significant growth engine, with countries like China and India exhibiting rapid market expansion due to a burgeoning young population, increasing healthcare expenditure, and improving access to antipyretic medicines. Regulatory support in these regions, focused on ensuring the availability of safe and affordable pediatric medications, further bolsters market growth. Investment trends in pharmaceutical research and development for pediatric formulations are notably high in these leading regions. For example, investment in R&D for novel Acetaminophen formulations in North America is projected to reach xx million dollars by 2025. Key drivers for dominance in these regions include extensive pediatrician networks that actively recommend specific antipyretics, robust pharmacovigilance systems that ensure product safety, and a strong consumer awareness regarding the importance of fever management in young children.
Antipyretic Medicine For Infants And Young Children Product Innovations
Product innovations in the Antipyretic Medicine for Infants and Young Children market are increasingly focused on enhancing safety, efficacy, and ease of administration for pediatric use. Novel formulations like orally disintegrating tablets and melt-in-your-mouth granules are gaining traction, offering a comfortable alternative for infants who struggle with liquid medications. Improved flavor profiles and reduced bitter aftertastes are also key selling points, enhancing patient compliance. Furthermore, advancements in controlled-release technologies are being explored to provide sustained fever relief and minimize the frequency of dosing. Manufacturers are also investigating the potential of combination products that offer synergistic effects for fever and mild pain relief, while meticulously adhering to stringent safety regulations for pediatric populations. The unique selling proposition of these innovations lies in their direct response to parental concerns for their children's well-being and comfort.
Propelling Factors for Antipyretic Medicine For Infants And Young Children Growth
Several key factors are propelling the growth of the Antipyretic Medicine for Infants and Young Children market. Rising global birth rates, particularly in emerging economies, directly translate to an increased demand for pediatric healthcare products. Growing parental awareness regarding the importance of timely and effective fever management, coupled with increased access to healthcare information and professional medical advice, significantly drives consumption. Technological advancements leading to improved drug delivery systems and more palatable formulations enhance product acceptance and compliance among young children. Furthermore, supportive government initiatives and favorable regulatory frameworks promoting the availability of safe and affordable antipyretic medications contribute to market expansion. The increasing prevalence of common childhood ailments necessitating fever reduction further solidifies demand.
Obstacles in the Antipyretic Medicine For Infants And Young Children Market
Despite robust growth, the Antipyretic Medicine for Infants and Young Children market faces several obstacles. Stringent regulatory hurdles and lengthy approval processes for new pediatric formulations can impede market entry and innovation. Supply chain disruptions, exacerbated by geopolitical factors and global health crises, can lead to product shortages and price volatility, impacting market stability. Intense competition among established players and the threat of counterfeit products can also affect market dynamics and consumer trust. Moreover, concerns regarding potential side effects and the risk of overdose, especially with incorrect dosing, necessitate continuous pharmacovigilance and consumer education. The economic impact of potential recalls, estimated to cost millions of dollars in lost revenue and brand damage, remains a significant deterrent.
Future Opportunities in Antipyretic Medicine For Infants And Young Children
Emerging opportunities in the Antipyretic Medicine for Infants and Young Children market are multifaceted. The burgeoning e-commerce sector presents a significant avenue for expanding reach and accessibility, particularly in remote areas. Development of novel, faster-acting formulations with extended efficacy periods offers a competitive edge. Furthermore, research into personalized medicine approaches, tailoring antipyretic treatments based on individual genetic predispositions or specific fever causes, holds immense future potential. Expanding into underserved emerging markets with growing pediatric populations and increasing healthcare expenditure also represents a substantial growth avenue. Innovations in sustainable packaging and eco-friendly manufacturing processes can also attract environmentally conscious consumers and stakeholders.
Major Players in the Antipyretic Medicine For Infants And Young Children Ecosystem
- Xinhua Pharmaceutical
- IOLCP
- Granules Biocause
- Strides Shasun
- Mallinckrodt
- Farmson
- Granules India
- SKPL
- Atabay
- Temad
- Anqiu Lu'an
- Zhejiang Kangle
- Hebei Jiheng
- Novacyl
- Anhui Fubore
- Anhui BBCA Likang
Key Developments in Antipyretic Medicine For Infants And Young Children Industry
- 2023, Q4: Xinhua Pharmaceutical launched a new, highly palatable Acetaminophen suspension formulation designed for infants, enhancing ease of administration and patient compliance.
- 2024, Q1: IOLCP announced a strategic partnership with a leading distributor to expand its reach in the Asian market, aiming to increase its market share by xx%.
- 2024, Q2: Granules India invested xx million dollars in expanding its manufacturing capacity for pediatric antipyretics, anticipating increased demand.
- 2024, Q3: Strides Shasun received regulatory approval for a novel Ibuprofen dosage form specifically for children aged 6 months to 2 years, a development valued at xx million dollars.
- 2024, Q4: Mallinckrodt reported positive results from clinical trials for a next-generation antipyretic agent with a potentially improved safety profile for infants, generating significant investor interest.
Strategic Antipyretic Medicine For Infants And Young Children Market Forecast
The Antipyretic Medicine for Infants and Young Children market is poised for sustained growth, driven by an increasing global birth rate and rising parental awareness regarding effective fever management. Key growth catalysts include ongoing innovation in child-friendly formulations, such as improved palatability and novel delivery systems, which directly address consumer demand for safer and more convenient options. The expanding reach of online sales channels and the increasing healthcare expenditure in emerging economies further bolster market potential. Strategic collaborations and targeted investments in research and development will be crucial for players to capture market share and introduce advanced therapeutic solutions, ensuring the continued availability of essential antipyretic medications for the pediatric population.
Antipyretic Medicine For Infants And Young Children Segmentation
-
1. Application
- 1.1. Online Sales
- 1.2. Offline Sales
-
2. Type
- 2.1. Acetaminophen
- 2.2. Ibuprofen
- 2.3. Other
Antipyretic Medicine For Infants And Young Children Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
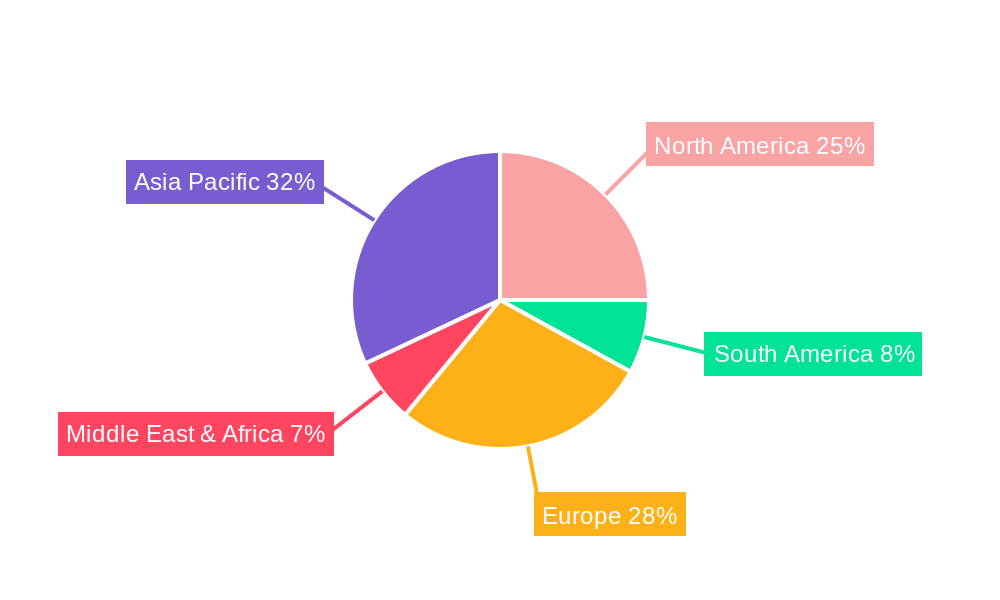
Antipyretic Medicine For Infants And Young Children Regional Market Share

Geographic Coverage of Antipyretic Medicine For Infants And Young Children
Antipyretic Medicine For Infants And Young Children REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XXX% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Antipyretic Medicine For Infants And Young Children Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Online Sales
- 5.1.2. Offline Sales
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Acetaminophen
- 5.2.2. Ibuprofen
- 5.2.3. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Antipyretic Medicine For Infants And Young Children Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Online Sales
- 6.1.2. Offline Sales
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Acetaminophen
- 6.2.2. Ibuprofen
- 6.2.3. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Antipyretic Medicine For Infants And Young Children Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Online Sales
- 7.1.2. Offline Sales
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Acetaminophen
- 7.2.2. Ibuprofen
- 7.2.3. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Antipyretic Medicine For Infants And Young Children Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Online Sales
- 8.1.2. Offline Sales
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Acetaminophen
- 8.2.2. Ibuprofen
- 8.2.3. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Antipyretic Medicine For Infants And Young Children Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Online Sales
- 9.1.2. Offline Sales
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Acetaminophen
- 9.2.2. Ibuprofen
- 9.2.3. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Antipyretic Medicine For Infants And Young Children Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Online Sales
- 10.1.2. Offline Sales
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Acetaminophen
- 10.2.2. Ibuprofen
- 10.2.3. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Xinhua Pharmaceutical
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 IOLCP
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Granules Biocause
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Strides Shasun
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Mallinckrodt
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Farmson
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Granules India
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 SKPL
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Atabay
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Temad
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Anqiu Lu'an
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Zhejiang Kangle
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Hebei Jiheng
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Novacyl
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Anhui Fubore
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Anhui BBCA Likang
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.1 Xinhua Pharmaceutical
List of Figures
- Figure 1: Global Antipyretic Medicine For Infants And Young Children Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Antipyretic Medicine For Infants And Young Children Revenue (million), by Application 2025 & 2033
- Figure 3: North America Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Antipyretic Medicine For Infants And Young Children Revenue (million), by Type 2025 & 2033
- Figure 5: North America Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Antipyretic Medicine For Infants And Young Children Revenue (million), by Country 2025 & 2033
- Figure 7: North America Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Antipyretic Medicine For Infants And Young Children Revenue (million), by Application 2025 & 2033
- Figure 9: South America Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Antipyretic Medicine For Infants And Young Children Revenue (million), by Type 2025 & 2033
- Figure 11: South America Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Antipyretic Medicine For Infants And Young Children Revenue (million), by Country 2025 & 2033
- Figure 13: South America Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Antipyretic Medicine For Infants And Young Children Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Antipyretic Medicine For Infants And Young Children Revenue (million), by Type 2025 & 2033
- Figure 17: Europe Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Antipyretic Medicine For Infants And Young Children Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global Antipyretic Medicine For Infants And Young Children Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Antipyretic Medicine For Infants And Young Children Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Antipyretic Medicine For Infants And Young Children?
The projected CAGR is approximately XXX%.
2. Which companies are prominent players in the Antipyretic Medicine For Infants And Young Children?
Key companies in the market include Xinhua Pharmaceutical, IOLCP, Granules Biocause, Strides Shasun, Mallinckrodt, Farmson, Granules India, SKPL, Atabay, Temad, Anqiu Lu'an, Zhejiang Kangle, Hebei Jiheng, Novacyl, Anhui Fubore, Anhui BBCA Likang.
3. What are the main segments of the Antipyretic Medicine For Infants And Young Children?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4250.00, USD 6375.00, and USD 8500.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Antipyretic Medicine For Infants And Young Children," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Antipyretic Medicine For Infants And Young Children report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Antipyretic Medicine For Infants And Young Children?
To stay informed about further developments, trends, and reports in the Antipyretic Medicine For Infants And Young Children, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


