Key Insights
The global clinical trial supply services market is experiencing robust growth, driven by an increasing number of clinical trials, the rising prevalence of chronic diseases, and the growing demand for advanced therapies. The market is characterized by a complex supply chain involving multiple stakeholders, including pharmaceutical companies, contract research organizations (CROs), and specialized logistics providers. Technological advancements, such as the adoption of blockchain technology for improved transparency and traceability, and the increasing use of digital tools for inventory management and supply chain optimization, are further fueling market expansion. While the market shows significant promise, challenges remain, including regulatory complexities, stringent quality control standards, and the need for efficient management of temperature-sensitive products. The market is segmented by service type (storage, packaging, distribution, labeling, etc.), therapeutic area (oncology, infectious diseases, etc.), and geographic region. Consolidation within the industry is anticipated as larger companies acquire smaller specialized firms to achieve economies of scale and expand their service offerings. The competitive landscape is characterized by both large multinational corporations and smaller specialized providers.
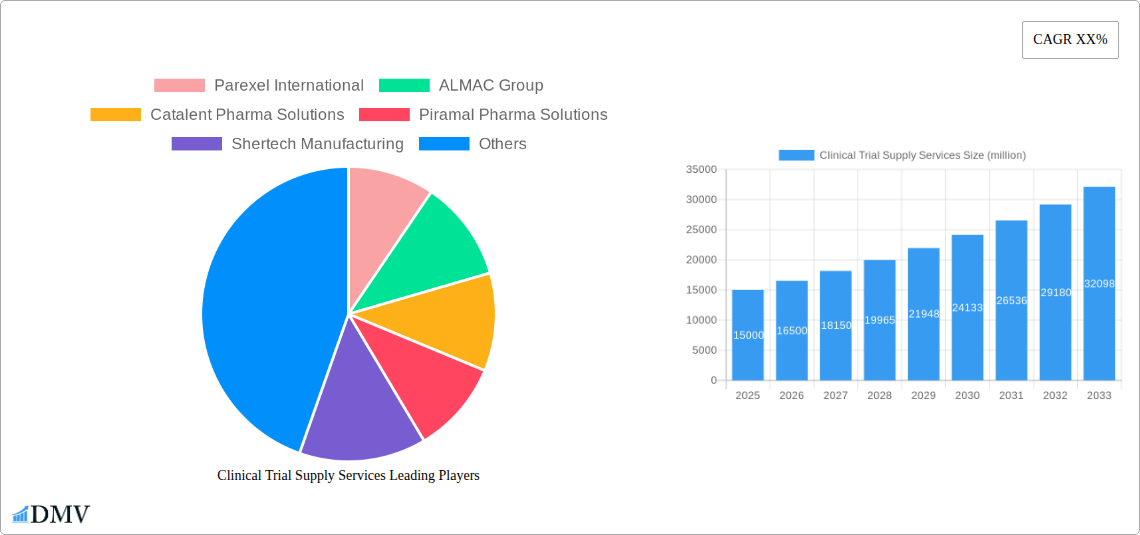
Clinical Trial Supply Services Market Size (In Billion)

The forecast period (2025-2033) anticipates continued growth, with a projected Compound Annual Growth Rate (CAGR) significantly influenced by factors such as the ongoing development of novel therapeutics, the expanding global clinical trials landscape, and the increasing adoption of innovative supply chain management techniques. Key players are strategically focusing on strengthening their infrastructure, expanding their geographic reach, and securing partnerships to gain a competitive edge. The market is anticipated to see substantial growth in emerging markets as healthcare infrastructure improves and clinical trial activity expands in these regions. However, maintaining stringent quality and regulatory compliance remains paramount, necessitating continuous investments in advanced technologies and robust quality control procedures to meet the stringent demands of this critical sector.
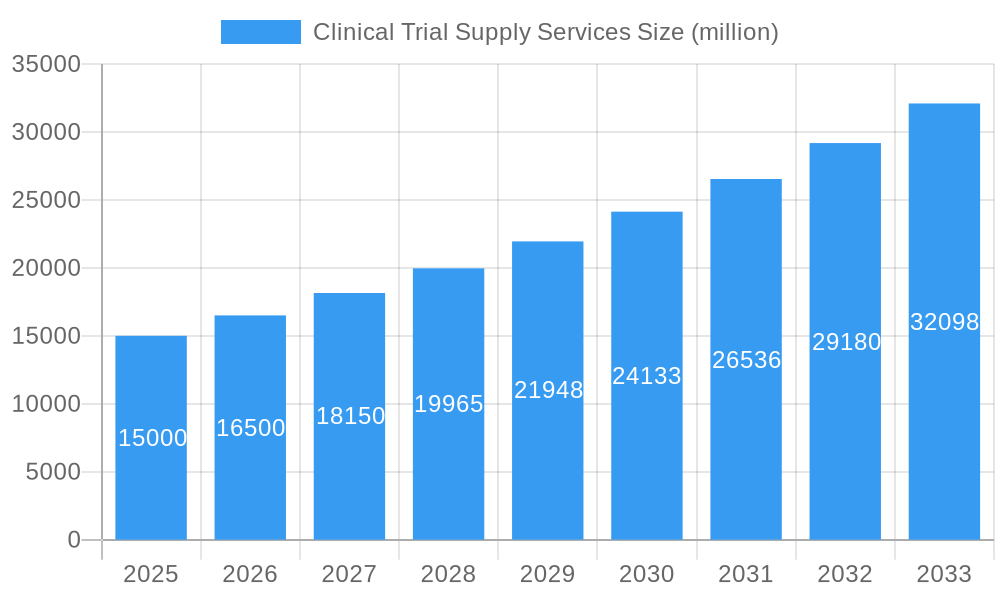
Clinical Trial Supply Services Company Market Share

Clinical Trial Supply Services Market Report: 2019-2033
This comprehensive report provides a detailed analysis of the Clinical Trial Supply Services market, projecting a market value exceeding $XX million by 2033. The study period spans 2019-2033, with 2025 serving as the base and estimated year. The forecast period is 2025-2033, and the historical period covers 2019-2024. Key players like Parexel International, ALMAC Group, Catalent Pharma Solutions, and others are analyzed for their market share, strategic moves, and future impact. This report is invaluable for stakeholders seeking to understand market dynamics, identify growth opportunities, and make informed strategic decisions.
Clinical Trial Supply Services Market Composition & Trends
The global Clinical Trial Supply Services market is experiencing robust growth, driven by increasing clinical trial activity and the rising complexity of drug development. Market concentration is moderate, with several major players holding significant shares, while numerous smaller companies cater to niche segments.
Market Share Distribution (Estimated 2025):
- Parexel International: XX%
- Catalent Pharma Solutions: XX%
- ALMAC Group: XX%
- Others: XX%
Innovation Catalysts: Advancements in cold chain technologies, automated packaging solutions, and innovative supply chain management systems are transforming the industry. Regulatory landscapes, particularly those concerning drug safety and data integrity, are continually evolving, presenting both challenges and opportunities. Substitute products are limited, primarily focusing on alternative storage and transportation methods. End-users, primarily pharmaceutical and biotech companies, are demanding greater efficiency, transparency, and cost-effectiveness from their supply chain partners. M&A activity has been significant, with deal values exceeding $XX million in recent years, indicating consolidation and a drive for scale. For example, the merger of Company A and Company B in 2022 resulted in a combined market share of XX%.
Clinical Trial Supply Services Industry Evolution
The Clinical Trial Supply Services market has witnessed substantial evolution over the past few years. From 2019 to 2024, the market exhibited a Compound Annual Growth Rate (CAGR) of XX%, fueled by several key factors. The increasing outsourcing of clinical trial supply chain management by pharmaceutical and biotechnology companies has significantly contributed to this growth. Technological advancements, such as the adoption of advanced analytics for inventory management and real-time tracking capabilities, have enhanced efficiency and reduced costs, further driving market expansion. Furthermore, the growing complexity of clinical trials, particularly those involving advanced therapies and personalized medicine, has increased the demand for specialized services. The shift towards decentralized clinical trials, which involves conducting trials outside of traditional clinical settings, has also created new opportunities and challenges. The adoption of blockchain technology and AI for inventory and logistics tracking is gaining traction, improving supply chain visibility and reducing waste. The market is expected to continue its upward trajectory, with a projected CAGR of XX% from 2025 to 2033, reaching a valuation exceeding $XX million. This growth will be fueled by continuous innovation, an increase in clinical trials for new therapies, and ongoing industry consolidation.
Leading Regions, Countries, or Segments in Clinical Trial Supply Services
North America currently holds the dominant position in the Clinical Trial Supply Services market, representing over XX% of the global market share in 2025.
- Key Drivers in North America:
- High concentration of pharmaceutical and biotechnology companies.
- Robust regulatory frameworks supporting clinical research.
- Significant investments in healthcare infrastructure and technology.
- Strong intellectual property protection.
The dominance of North America stems from a combination of factors, including the high concentration of pharmaceutical and biotechnology companies, well-established regulatory frameworks, substantial investments in healthcare infrastructure and advanced technology, and a strong intellectual property protection regime. Europe and Asia-Pacific are also significant markets, witnessing considerable growth driven by increasing healthcare spending and clinical trial activity. However, regulatory hurdles and infrastructure limitations in certain regions continue to pose challenges to market expansion.
Clinical Trial Supply Services Product Innovations
Recent years have witnessed a surge in product innovations within the Clinical Trial Supply Services sector. Smart packaging solutions incorporating real-time temperature monitoring and data logging are gaining traction, ensuring the integrity of temperature-sensitive drug products throughout the supply chain. Automated dispensing systems and robotic process automation are enhancing efficiency and reducing human error in kitting and packaging processes. These advancements are not only improving the efficiency and accuracy of clinical trial supply chains but are also enhancing drug product quality and patient safety. The adoption of advanced analytics allows for proactive identification of potential supply chain disruptions and improved inventory management, leading to significant cost savings and better decision-making.
Propelling Factors for Clinical Trial Supply Services Growth
Several factors are driving the robust growth of the Clinical Trial Supply Services market. Firstly, the ever-increasing number of clinical trials being conducted globally due to an expanding pipeline of new drugs and therapies fuels demand for specialized services. Secondly, technological advancements are leading to more efficient and cost-effective solutions in areas such as cold chain management and inventory tracking. Finally, favorable regulatory environments in several key markets are encouraging investments and innovation within the industry.
Obstacles in the Clinical Trial Supply Services Market
Despite the positive growth outlook, the Clinical Trial Supply Services market faces several significant challenges. Strict regulatory requirements and compliance standards can increase costs and complexity for providers. Supply chain disruptions, particularly those related to geopolitical events and natural disasters, pose a considerable risk to the timely delivery of critical clinical trial supplies. Intense competition from both established players and new entrants is putting pressure on pricing and profit margins. These challenges necessitate a robust and adaptable approach to managing risks and seizing opportunities effectively.
Future Opportunities in Clinical Trial Supply Services
The future of Clinical Trial Supply Services presents exciting opportunities. The growth of decentralized clinical trials will create a demand for innovative supply chain solutions catering to diverse geographical locations and trial settings. The integration of advanced technologies like artificial intelligence and blockchain will provide greater transparency, efficiency, and security. Expansion into emerging markets with growing healthcare sectors presents significant untapped potential for growth. Focusing on personalized medicine and advanced therapies will necessitate customized supply chain strategies, creating additional service offerings.
Major Players in the Clinical Trial Supply Services Ecosystem
- Parexel International
- ALMAC Group
- Catalent Pharma Solutions
- Piramal Pharma Solutions
- Shertech Manufacturing
- Thermo Fisher Scientific
- PCI Services
- Patheon, Inc.
- Sharp Packaging Services
- Biocair
- Movianto
- Klifo A/S
Key Developments in Clinical Trial Supply Services Industry
- 2022, Q4: Catalent Pharma Solutions announced a significant investment in expanding its cold chain capabilities.
- 2023, Q1: ALMAC Group launched a new, automated packaging solution for clinical trial materials.
- 2023, Q3: Parexel International acquired a smaller competitor, strengthening its market position. (Further details on acquisitions and mergers are included in the full report)
Strategic Clinical Trial Supply Services Market Forecast
The Clinical Trial Supply Services market is poised for continued robust growth, driven by the ongoing expansion of clinical trials and technological advancements. The increasing demand for personalized medicine and advanced therapies will create new opportunities for specialized supply chain solutions. Strategic partnerships, investments in automation, and expansion into emerging markets will be crucial for companies seeking to thrive in this dynamic landscape. The market is expected to exceed $XX million by 2033, presenting substantial opportunities for both established players and new entrants.
Clinical Trial Supply Services Segmentation
-
1. Application
- 1.1. Oncology
- 1.2. Cardiovascular Diseases
- 1.3. Respiratory Diseases
- 1.4. CNS And Mental Disorders
- 1.5. Dermatology
- 1.6. Blood Disorders
- 1.7. Infectious Diseases
- 1.8. Others
-
2. Types
- 2.1. Manufacturing
- 2.2. Packaging and Labeling
- 2.3. Logistics and Distribution
Clinical Trial Supply Services Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
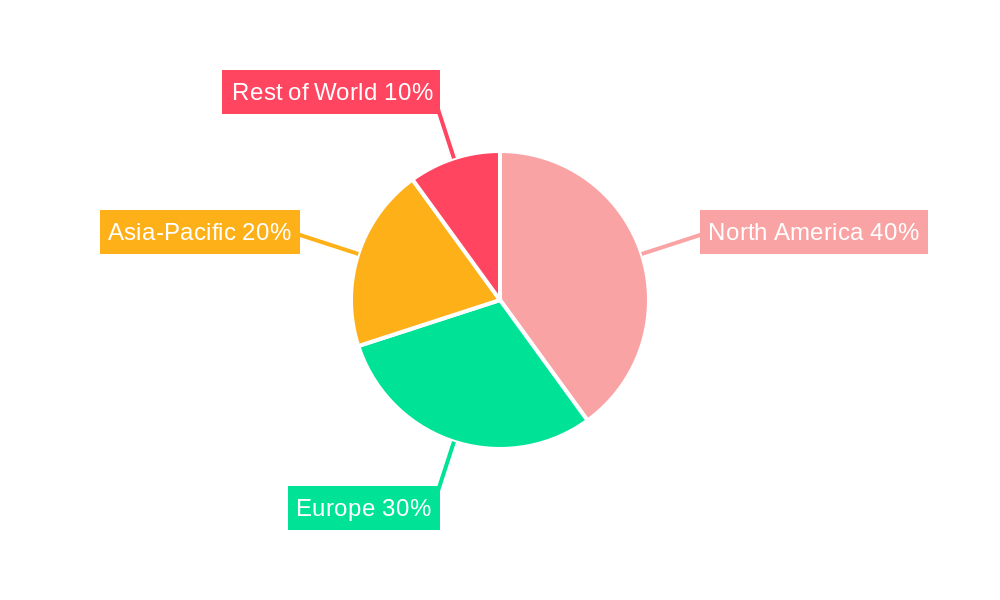
Clinical Trial Supply Services Regional Market Share

Geographic Coverage of Clinical Trial Supply Services
Clinical Trial Supply Services REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Clinical Trial Supply Services Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Oncology
- 5.1.2. Cardiovascular Diseases
- 5.1.3. Respiratory Diseases
- 5.1.4. CNS And Mental Disorders
- 5.1.5. Dermatology
- 5.1.6. Blood Disorders
- 5.1.7. Infectious Diseases
- 5.1.8. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Manufacturing
- 5.2.2. Packaging and Labeling
- 5.2.3. Logistics and Distribution
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Clinical Trial Supply Services Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Oncology
- 6.1.2. Cardiovascular Diseases
- 6.1.3. Respiratory Diseases
- 6.1.4. CNS And Mental Disorders
- 6.1.5. Dermatology
- 6.1.6. Blood Disorders
- 6.1.7. Infectious Diseases
- 6.1.8. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Manufacturing
- 6.2.2. Packaging and Labeling
- 6.2.3. Logistics and Distribution
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Clinical Trial Supply Services Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Oncology
- 7.1.2. Cardiovascular Diseases
- 7.1.3. Respiratory Diseases
- 7.1.4. CNS And Mental Disorders
- 7.1.5. Dermatology
- 7.1.6. Blood Disorders
- 7.1.7. Infectious Diseases
- 7.1.8. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Manufacturing
- 7.2.2. Packaging and Labeling
- 7.2.3. Logistics and Distribution
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Clinical Trial Supply Services Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Oncology
- 8.1.2. Cardiovascular Diseases
- 8.1.3. Respiratory Diseases
- 8.1.4. CNS And Mental Disorders
- 8.1.5. Dermatology
- 8.1.6. Blood Disorders
- 8.1.7. Infectious Diseases
- 8.1.8. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Manufacturing
- 8.2.2. Packaging and Labeling
- 8.2.3. Logistics and Distribution
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Clinical Trial Supply Services Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Oncology
- 9.1.2. Cardiovascular Diseases
- 9.1.3. Respiratory Diseases
- 9.1.4. CNS And Mental Disorders
- 9.1.5. Dermatology
- 9.1.6. Blood Disorders
- 9.1.7. Infectious Diseases
- 9.1.8. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Manufacturing
- 9.2.2. Packaging and Labeling
- 9.2.3. Logistics and Distribution
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Clinical Trial Supply Services Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Oncology
- 10.1.2. Cardiovascular Diseases
- 10.1.3. Respiratory Diseases
- 10.1.4. CNS And Mental Disorders
- 10.1.5. Dermatology
- 10.1.6. Blood Disorders
- 10.1.7. Infectious Diseases
- 10.1.8. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Manufacturing
- 10.2.2. Packaging and Labeling
- 10.2.3. Logistics and Distribution
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Parexel International
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 ALMAC Group
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Catalent Pharma Solutions
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Piramal Pharma Solutions
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Shertech Manufacturing
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Thermo Fisher Scientific
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 PCI Services
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Patheon
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Inc.
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Sharp Packaging Services
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Biocair
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Movianto
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Klifo A/S.
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.1 Parexel International
List of Figures
- Figure 1: Global Clinical Trial Supply Services Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Clinical Trial Supply Services Revenue (million), by Application 2025 & 2033
- Figure 3: North America Clinical Trial Supply Services Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Clinical Trial Supply Services Revenue (million), by Types 2025 & 2033
- Figure 5: North America Clinical Trial Supply Services Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Clinical Trial Supply Services Revenue (million), by Country 2025 & 2033
- Figure 7: North America Clinical Trial Supply Services Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Clinical Trial Supply Services Revenue (million), by Application 2025 & 2033
- Figure 9: South America Clinical Trial Supply Services Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Clinical Trial Supply Services Revenue (million), by Types 2025 & 2033
- Figure 11: South America Clinical Trial Supply Services Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Clinical Trial Supply Services Revenue (million), by Country 2025 & 2033
- Figure 13: South America Clinical Trial Supply Services Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Clinical Trial Supply Services Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Clinical Trial Supply Services Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Clinical Trial Supply Services Revenue (million), by Types 2025 & 2033
- Figure 17: Europe Clinical Trial Supply Services Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Clinical Trial Supply Services Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Clinical Trial Supply Services Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Clinical Trial Supply Services Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Clinical Trial Supply Services Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Clinical Trial Supply Services Revenue (million), by Types 2025 & 2033
- Figure 23: Middle East & Africa Clinical Trial Supply Services Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Clinical Trial Supply Services Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Clinical Trial Supply Services Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Clinical Trial Supply Services Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Clinical Trial Supply Services Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Clinical Trial Supply Services Revenue (million), by Types 2025 & 2033
- Figure 29: Asia Pacific Clinical Trial Supply Services Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Clinical Trial Supply Services Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Clinical Trial Supply Services Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Clinical Trial Supply Services Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Clinical Trial Supply Services Revenue million Forecast, by Types 2020 & 2033
- Table 3: Global Clinical Trial Supply Services Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Clinical Trial Supply Services Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Clinical Trial Supply Services Revenue million Forecast, by Types 2020 & 2033
- Table 6: Global Clinical Trial Supply Services Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Clinical Trial Supply Services Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Clinical Trial Supply Services Revenue million Forecast, by Types 2020 & 2033
- Table 12: Global Clinical Trial Supply Services Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Clinical Trial Supply Services Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Clinical Trial Supply Services Revenue million Forecast, by Types 2020 & 2033
- Table 18: Global Clinical Trial Supply Services Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Clinical Trial Supply Services Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Clinical Trial Supply Services Revenue million Forecast, by Types 2020 & 2033
- Table 30: Global Clinical Trial Supply Services Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Clinical Trial Supply Services Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Clinical Trial Supply Services Revenue million Forecast, by Types 2020 & 2033
- Table 39: Global Clinical Trial Supply Services Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Clinical Trial Supply Services Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Clinical Trial Supply Services?
The projected CAGR is approximately XX%.
2. Which companies are prominent players in the Clinical Trial Supply Services?
Key companies in the market include Parexel International, ALMAC Group, Catalent Pharma Solutions, Piramal Pharma Solutions, Shertech Manufacturing, Thermo Fisher Scientific, PCI Services, Patheon, Inc., Sharp Packaging Services, Biocair, Movianto, Klifo A/S..
3. What are the main segments of the Clinical Trial Supply Services?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 5600.00, USD 8400.00, and USD 11200.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Clinical Trial Supply Services," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Clinical Trial Supply Services report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Clinical Trial Supply Services?
To stay informed about further developments, trends, and reports in the Clinical Trial Supply Services, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


