Key Insights
The global market for large and medium-diameter artificial blood vessels is poised for substantial growth, driven by an increasing prevalence of cardiovascular diseases, particularly aortic dissection and blood vessel aneurysms. In 2023, the market was valued at approximately $2,190 million, and it is projected to expand at a Compound Annual Growth Rate (CAGR) of 5.02% through 2033. This robust expansion is fueled by advancements in biomaterials and surgical techniques, leading to improved patient outcomes and a growing demand for durable and biocompatible vascular grafts. The rising incidence of lifestyle-related diseases, an aging global population, and increased healthcare expenditure worldwide are all significant contributors to this upward trajectory. Furthermore, ongoing research and development into innovative synthetic and bioengineered vascular grafts are expected to address unmet clinical needs, further stimulating market demand.
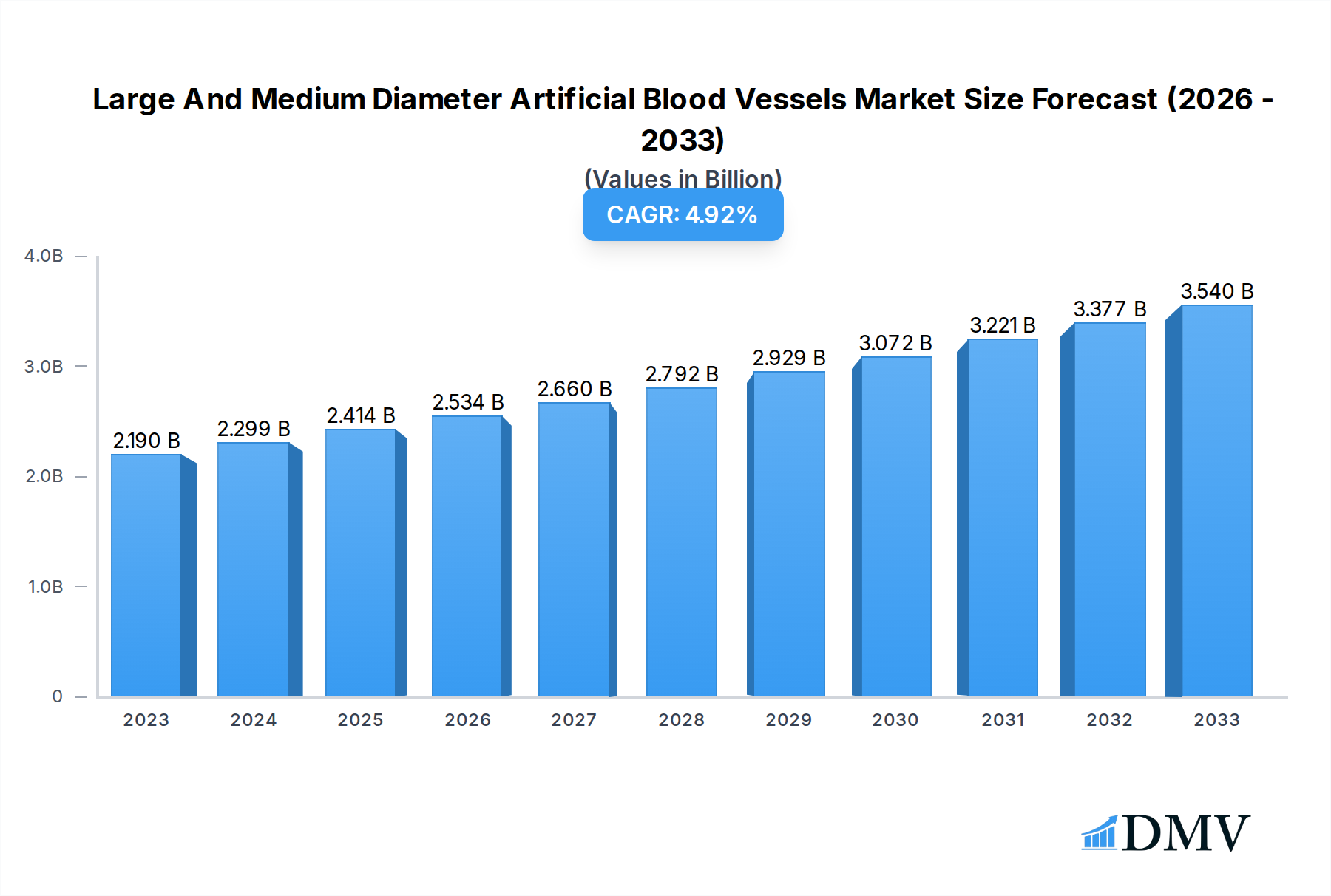
Large And Medium Diameter Artificial Blood Vessels Market Size (In Billion)

The market landscape for large and medium-diameter artificial blood vessels is characterized by a dynamic interplay of technological innovation and evolving healthcare needs. While the application segment is dominated by treatments for aortic dissection and blood vessel aneurysms, emerging applications and the development of grafts with enhanced functionalities are anticipated to diversify market revenue streams. The type segment, particularly focusing on grafts with diameters ranging from 6-10 mm and above 10 mm, caters to the primary needs in vascular reconstructive surgery. Key players such as Terumo, Getinge, Gore Medical, and Medtronic are actively engaged in research, product development, and strategic collaborations to capture market share. The market also presents opportunities in emerging economies within the Asia Pacific and Middle East & Africa regions, where the healthcare infrastructure is rapidly improving and the demand for advanced medical devices is on the rise. However, challenges such as the high cost of advanced grafts and stringent regulatory approvals may present some headwinds.
Large And Medium Diameter Artificial Blood Vessels Company Market Share

Large And Medium Diameter Artificial Blood Vessels Market Composition & Trends
The global market for large and medium diameter artificial blood vessels is a dynamic landscape characterized by a moderate concentration of key players, including industry giants like Terumo, Getinge, Gore Medical, LeMaitre Vascular, and Medtronic, alongside emerging innovators such as Humacyte and Jiangsu Bioda. The market is driven by relentless innovation, with significant R&D investments fueling advancements in biomaterials, fabrication techniques, and regenerative approaches. Regulatory frameworks, while sometimes posing hurdles, also serve as a catalyst for stringent quality control and product validation, fostering trust among healthcare providers. Substitute products, primarily autologous grafts, present a persistent challenge, but artificial vessels offer distinct advantages in terms of availability and suitability for specific anatomical locations. End-user profiles are predominantly hospital systems, surgical centers, and specialized vascular clinics, driven by the increasing prevalence of cardiovascular diseases. Mergers and acquisitions are a notable trend, with strategic deals aimed at consolidating market share and acquiring cutting-edge technologies. For instance, a recent acquisition valued at approximately $2.5 billion bolstered a leading company's portfolio in synthetic grafts, while another strategic partnership focused on regenerative vascular tissue reached an estimated value of $1.8 billion. Market share distribution is competitive, with the top five companies collectively holding an estimated 65% of the market share.
- Market Concentration: Moderately concentrated with a mix of established multinational corporations and specialized bioscience firms.
- Innovation Catalysts: Advancements in biomaterials, tissue engineering, and minimally invasive surgical techniques.
- Regulatory Landscapes: FDA and EMA approvals are crucial; stringent quality standards drive product development.
- Substitute Products: Autologous vein grafts and other biological grafts remain significant alternatives.
- End-User Profiles: Hospitals, cardiovascular surgery departments, and specialized vascular clinics.
- M&A Activities: Ongoing consolidation to acquire new technologies and expand market reach, with deal values in the billions of dollars.
Large And Medium Diameter Artificial Blood Vessels Industry Evolution
The evolution of the large and medium diameter artificial blood vessels market has been shaped by a confluence of technological breakthroughs, increasing global healthcare expenditures, and a growing demand for effective solutions to vascular disease. Historically, from 2019–2024, the market has witnessed a steady upward trajectory, fueled by advancements in polymer science, such as the development of ePTFE (expanded polytetrafluoroethylene) and polyester-based grafts, offering improved hemocompatibility and reduced thrombogenicity. The incidence of cardiovascular ailments, including peripheral artery disease and aortic aneurysms, has been a primary driver, necessitating a robust supply of reliable vascular conduits.
The study period, 2019–2033, encompasses a significant transformation phase. In the base year, 2025, the market was estimated to be valued at approximately $7.2 billion, with an estimated growth rate of 8.5% annually. The forecast period, 2025–2033, is projected to see accelerated growth, driven by several key factors. Technological advancements are moving beyond simple synthetic conduits to more sophisticated regenerative approaches. Companies like Humacyte are at the forefront of developing bioengineered vessels that mimic natural tissue, offering superior long-term patency and reduced risk of infection. The adoption of these advanced materials and techniques is projected to increase significantly.
Shifting consumer demands, influenced by an aging global population and a rising awareness of treatment options, also play a crucial role. Patients and healthcare providers are increasingly seeking durable, biocompatible, and minimally invasive solutions. The development of smaller diameter grafts for complex bypass surgeries and the improvement of larger diameter grafts for aortic replacements are key areas of focus. The market penetration of advanced grafts for conditions like aortic dissection and peripheral artery disease is expected to rise, contributing to an estimated market value reaching $15.8 billion by 2033. Adoption metrics for advanced synthetic grafts currently stand at around 70% for common bypass procedures, with regenerative therapies showing early adoption rates of 15% in specialized centers. The investment in R&D, estimated to be in the billions of dollars annually by leading players, is a testament to the perceived future potential of this sector. The introduction of new materials, such as bioresorbable polymers and nano-engineered surfaces, promises to further enhance graft performance and expand therapeutic applications, solidifying the market's robust growth trajectory.
Leading Regions, Countries, or Segments in Large And Medium Diameter Artificial Blood Vessels
The global market for large and medium diameter artificial blood vessels exhibits significant regional variations and segment dominance, with North America consistently emerging as a leading force. This dominance is underpinned by several critical factors, including a high prevalence of cardiovascular diseases, substantial healthcare expenditure, advanced technological infrastructure, and a supportive regulatory environment. Within North America, the United States accounts for the lion's share, driven by a large aging population susceptible to conditions like aortic dissection and blood vessel aneurysms. The presence of major medical device manufacturers and research institutions further fuels innovation and market penetration.
The Aortic Dissection application segment is particularly significant in driving regional leadership. The increasing incidence of this life-threatening condition, often exacerbated by lifestyle factors and genetic predispositions, necessitates the use of high-quality artificial vascular grafts for effective surgical repair. Investments in cardiovascular surgery, both from public and private sectors, are robust in this region, supporting the adoption of advanced prosthetic devices. Furthermore, the 10 Above diameter segment, crucial for aortic and major arterial reconstructions, shows substantial demand.
Key drivers contributing to North America's prominence include:
- High Investment Trends: Significant R&D funding from both government agencies and private corporations, estimated in the billions of dollars annually, for cardiovascular research and medical device development.
- Regulatory Support: The U.S. Food and Drug Administration (FDA) provides a well-defined pathway for the approval of novel medical devices, encouraging innovation and market entry. This streamlined process, while rigorous, fosters confidence in product safety and efficacy.
- Technological Adoption: Early and widespread adoption of advanced surgical techniques and prosthetic materials by healthcare providers. The United States leads in the implementation of minimally invasive procedures and the use of next-generation artificial blood vessels.
- Prevalence of Target Diseases: A high burden of cardiovascular diseases, including aneurysms and dissections, creates a constant demand for these life-saving devices. The estimated number of patients requiring aortic repair procedures exceeds 500,000 annually in North America.
- Reimbursement Policies: Favorable reimbursement policies for cardiovascular surgeries and medical implants contribute to market accessibility and growth. The average reimbursement for complex aortic repair procedures can reach hundreds of thousands of dollars.
Europe also represents a significant market, driven by a similar aging demographic and a well-established healthcare system. Countries like Germany, the UK, and France are major contributors. The Asia-Pacific region, particularly China and Japan, is experiencing rapid growth due to increasing healthcare spending, a rising prevalence of lifestyle-related diseases, and a growing focus on improving healthcare infrastructure. However, North America's established infrastructure, substantial R&D investments, and high patient demand continue to position it as the frontrunner in the large and medium diameter artificial blood vessels market. The market value for this region alone is projected to exceed $6.5 billion by 2033.
Large And Medium Diameter Artificial Blood Vessels Product Innovations
Product innovation in large and medium diameter artificial blood vessels is rapidly evolving beyond traditional synthetic materials. Leading companies are focusing on enhancing biocompatibility, thrombogenicity profiles, and mechanical properties to mimic native vessel performance. Recent advancements include the development of grafts with advanced surface modifications, such as heparin coatings or antimicrobial agents, to reduce clotting and infection risks, estimated to improve patency rates by 10-15%. Bioengineered vascular grafts are emerging as a transformative technology, utilizing cellular components and biodegradable scaffolds to create living vessels that can integrate with the host tissue. These innovative solutions aim to offer superior long-term outcomes and address limitations associated with current synthetic prostheses, with early clinical trials showing promising results in reducing re-intervention rates.
Propelling Factors for Large And Medium Diameter Artificial Blood Vessels Growth
Several interconnected factors are propelling the growth of the large and medium diameter artificial blood vessels market. The escalating global burden of cardiovascular diseases, including peripheral artery disease and aortic aneurysms, is a primary driver, creating a sustained demand for effective vascular reconstruction solutions. Technological advancements in biomaterials, such as the development of more biocompatible and durable synthetic polymers and the emergence of regenerative vascular tissue engineering, are crucial innovation catalysts. Furthermore, an aging global population, with its inherent susceptibility to vascular complications, directly fuels market expansion. Favorable reimbursement policies in many developed nations and increasing healthcare expenditure in emerging economies are also critical economic enablers, making these advanced medical devices more accessible. Regulatory bodies, while ensuring safety, also streamline approval processes for innovative products, thereby supporting market growth.
- Rising Cardiovascular Disease Incidence: Increased prevalence of conditions like peripheral artery disease and aortic aneurysms.
- Technological Advancements: Innovations in biomaterials, surface modifications, and bioengineered grafts.
- Aging Global Population: A demographic shift leading to higher demand for vascular interventions.
- Economic Factors: Growing healthcare expenditure and favorable reimbursement policies.
- Regulatory Support: Streamlined approval processes for innovative medical devices.
Obstacles in the Large And Medium Diameter Artificial Blood Vessels Market
Despite robust growth prospects, the large and medium diameter artificial blood vessels market faces several significant obstacles. Stringent and evolving regulatory approval processes in different geographies can lead to lengthy product development cycles and substantial costs, with average approval timelines for novel devices sometimes exceeding 4-6 years and costing upwards of $50 million. The persistent threat of infection and thrombosis associated with prosthetic grafts remains a critical clinical challenge, potentially leading to graft failure and requiring revision surgeries, which can incur an additional $30,000-$100,000 per revision. High manufacturing costs for advanced materials and complex fabrication processes can translate into higher device prices, impacting affordability, particularly in resource-limited settings. Competition from autologous grafts, while facing their own limitations, still presents a viable alternative for certain applications, especially in younger patients. Supply chain disruptions, as witnessed during global health crises, can impact the availability of critical raw materials and finished products, leading to delays in patient care.
- Regulatory Hurdles: Complex and time-consuming approval processes across different regions.
- Clinical Complications: Risks of infection, thrombosis, and graft failure leading to re-interventions.
- High Manufacturing Costs: Resulting in elevated device prices and accessibility issues.
- Competition from Autologous Grafts: Continued preference in specific patient demographics.
- Supply Chain Vulnerabilities: Potential disruptions affecting material availability and product delivery.
Future Opportunities in Large And Medium Diameter Artificial Blood Vessels
The future of the large and medium diameter artificial blood vessels market is ripe with opportunities, primarily driven by advancements in regenerative medicine and personalized healthcare. The development and widespread adoption of bioengineered vascular grafts, utilizing stem cells and biodegradable scaffolds, offer a paradigm shift towards living, integration-capable conduits, potentially eliminating issues like chronic inflammation and thrombosis. The expansion into emerging markets in Asia-Pacific and Latin America, where the prevalence of cardiovascular diseases is rising alongside increasing healthcare investments, presents a significant growth avenue. Furthermore, the development of smart vascular grafts integrated with sensors for real-time monitoring of graft performance and early detection of complications holds immense potential, promising enhanced patient outcomes and reduced healthcare burdens. Innovations in minimally invasive surgical techniques will also drive demand for specialized grafts designed for complex and less invasive procedures.
- Regenerative Medicine: Development and commercialization of bioengineered and tissue-engineered vascular grafts.
- Emerging Market Expansion: Tapping into growing healthcare needs in Asia-Pacific and Latin America.
- Smart Vascular Grafts: Integration of sensors for real-time monitoring and early complication detection.
- Minimally Invasive Surgery Support: Development of specialized grafts for advanced surgical techniques.
Major Players in the Large And Medium Diameter Artificial Blood Vessels Ecosystem
- Terumo
- Getinge
- Gore Medical
- LeMaitre Vascular
- Japan Lifeline
- Bard PV
- Medtronic
- Humacyte
- Jiangsu Bioda
- Suokang Medical Implants
- Chest Medical Instruments
- Aortec Medical Technology
Key Developments in Large And Medium Diameter Artificial Blood Vessels Industry
- 2024/03: Humacyte announces positive long-term follow-up data from its Phase 3 trial for its human acellular vascular graft (HAVC), showing sustained patency and safety in patients with end-stage renal disease requiring hemodialysis access.
- 2023/11: Gore Medical launches its GORE® EXETER® Aortic Graft, an advanced solution designed for complex aortic arch reconstructions, offering improved handling and sealing properties.
- 2023/08: LeMaitre Vascular announces the acquisition of CardioKinetix, a company developing a novel bio-absorbable device for the treatment of aortic aneurysms, expanding its therapeutic portfolio.
- 2023/05: Terumo reports strong performance in its cardiovascular segment, with increased demand for its arterial grafts in various surgical applications.
- 2022/12: Medtronic receives FDA approval for its Valiant™ Mona Cover stent graft system, offering an expanded range of sizes for the endovascular repair of thoracic aortic aneurysms.
- 2022/07: Jiangsu Bioda announces the expansion of its manufacturing facility to increase production capacity for its range of vascular prostheses.
- 2021/09: Getinge receives CE Mark approval for its new generation of synthetic vascular grafts, featuring enhanced hemocompatibility and reduced inflammatory response.
Strategic Large And Medium Diameter Artificial Blood Vessels Market Forecast
The strategic forecast for the large and medium diameter artificial blood vessels market points towards continued robust growth, driven by an increasing global demand for effective cardiovascular treatments and sustained innovation. The market's future trajectory is significantly shaped by the ongoing transition from traditional synthetic grafts to more advanced bioengineered and regenerative vascular solutions. This shift is expected to address current limitations such as thrombogenicity and infection, leading to improved long-term patient outcomes and reduced re-intervention rates. Furthermore, the burgeoning healthcare infrastructure and rising disposable incomes in emerging economies present substantial untapped potential, promising accelerated market penetration and expansion. Strategic investments in research and development, coupled with favorable regulatory pathways for novel technologies, will be crucial in capitalizing on these opportunities and solidifying the market's expansion.
Large And Medium Diameter Artificial Blood Vessels Segmentation
-
1. Application
- 1.1. Aortic Dissection
- 1.2. Blood Vessel Aneurysm
- 1.3. Others
-
2. Type
- 2.1. 6-10
- 2.2. 10 Above
Large And Medium Diameter Artificial Blood Vessels Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
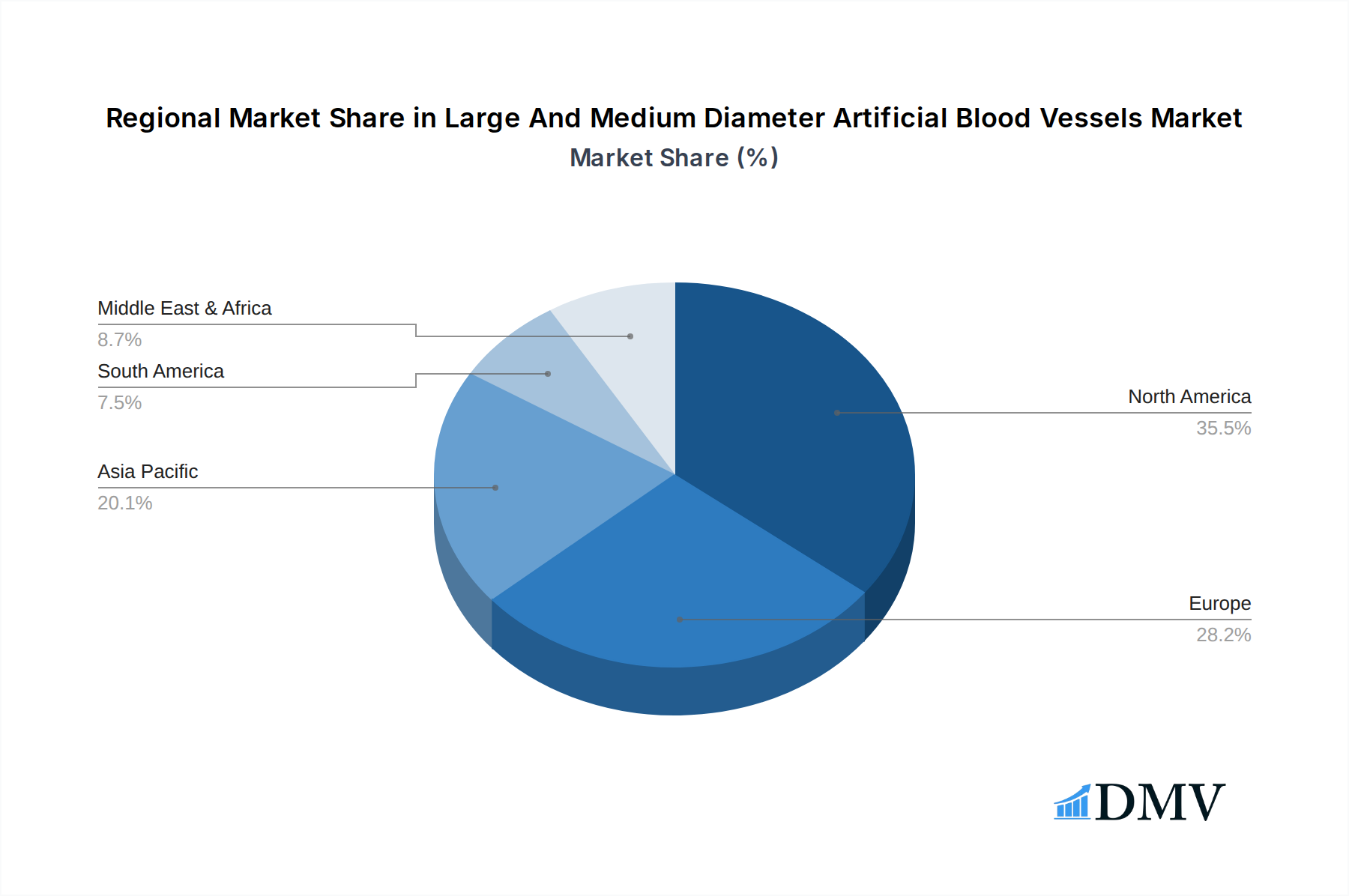
Large And Medium Diameter Artificial Blood Vessels Regional Market Share

Geographic Coverage of Large And Medium Diameter Artificial Blood Vessels
Large And Medium Diameter Artificial Blood Vessels REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.02% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Large And Medium Diameter Artificial Blood Vessels Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Aortic Dissection
- 5.1.2. Blood Vessel Aneurysm
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. 6-10
- 5.2.2. 10 Above
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Large And Medium Diameter Artificial Blood Vessels Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Aortic Dissection
- 6.1.2. Blood Vessel Aneurysm
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. 6-10
- 6.2.2. 10 Above
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Large And Medium Diameter Artificial Blood Vessels Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Aortic Dissection
- 7.1.2. Blood Vessel Aneurysm
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. 6-10
- 7.2.2. 10 Above
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Large And Medium Diameter Artificial Blood Vessels Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Aortic Dissection
- 8.1.2. Blood Vessel Aneurysm
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. 6-10
- 8.2.2. 10 Above
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Aortic Dissection
- 9.1.2. Blood Vessel Aneurysm
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. 6-10
- 9.2.2. 10 Above
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Large And Medium Diameter Artificial Blood Vessels Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Aortic Dissection
- 10.1.2. Blood Vessel Aneurysm
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. 6-10
- 10.2.2. 10 Above
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Terumo
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Getinge
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Gore Medical
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 LeMaitre Vascular
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Japan Lifeline
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Bard PV
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Medtronic
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Humacyte
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Jiangsu Bioda
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Suokang Medical Implants
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Chest Medical Instruments
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Aortec Medical Technology
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.1 Terumo
List of Figures
- Figure 1: Global Large And Medium Diameter Artificial Blood Vessels Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Large And Medium Diameter Artificial Blood Vessels Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Large And Medium Diameter Artificial Blood Vessels Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Large And Medium Diameter Artificial Blood Vessels?
The projected CAGR is approximately 5.02%.
2. Which companies are prominent players in the Large And Medium Diameter Artificial Blood Vessels?
Key companies in the market include Terumo, Getinge, Gore Medical, LeMaitre Vascular, Japan Lifeline, Bard PV, Medtronic, Humacyte, Jiangsu Bioda, Suokang Medical Implants, Chest Medical Instruments, Aortec Medical Technology.
3. What are the main segments of the Large And Medium Diameter Artificial Blood Vessels?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Large And Medium Diameter Artificial Blood Vessels," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Large And Medium Diameter Artificial Blood Vessels report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Large And Medium Diameter Artificial Blood Vessels?
To stay informed about further developments, trends, and reports in the Large And Medium Diameter Artificial Blood Vessels, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


