Key Insights
The global Intravenous Iron Drugs market is poised for significant expansion, with an estimated market size of USD 3.6 billion in 2025. This growth is propelled by a robust Compound Annual Growth Rate (CAGR) of 9.6% projected over the forecast period of 2025-2033. Key drivers fueling this upward trajectory include the increasing prevalence of chronic diseases like Chronic Kidney Disease (CKD), Irritable Bowel Disease (IBD), and various forms of cancer, all of which necessitate effective iron deficiency management. Advances in drug delivery systems and the development of novel iron formulations offering improved safety and efficacy are also contributing to market dynamism. The growing awareness among healthcare professionals and patients regarding the benefits of intravenous iron therapy for rapid replenishment of iron stores, particularly in cases where oral supplements are insufficient or poorly tolerated, is a crucial factor. Furthermore, expanding healthcare infrastructure and increased access to advanced medical treatments in emerging economies are expected to create new avenues for market growth.
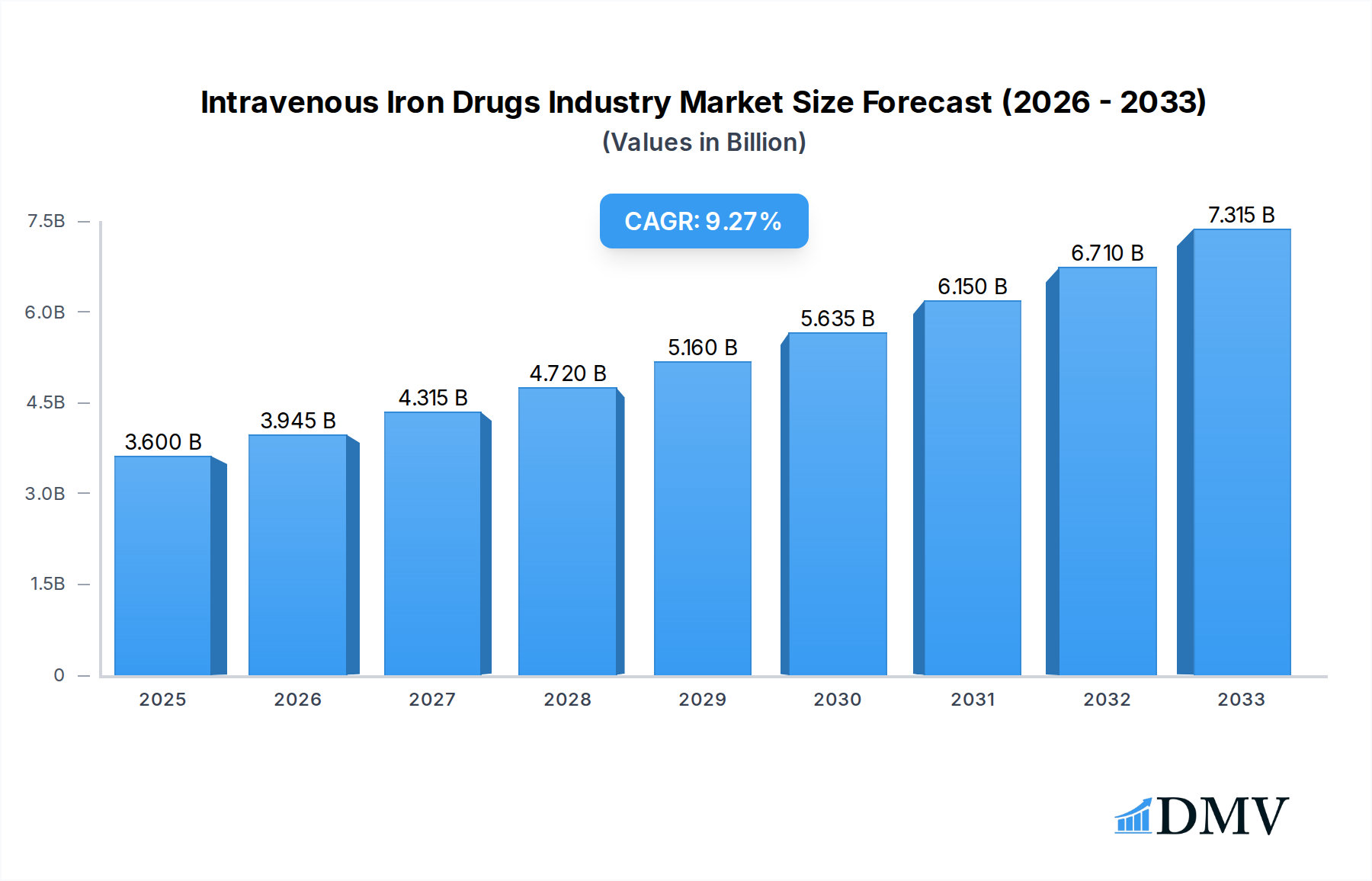
Intravenous Iron Drugs Industry Market Size (In Billion)

The market is segmented across various product types, including Ferric Carboxymaltose, Iron Sucrose, and Iron Dextran, each catering to specific clinical needs and patient profiles. The application segment is dominated by Chronic Kidney Disease, reflecting the high demand for iron therapy in this patient population. However, significant growth is also anticipated in the application areas of IBD and cancer, driven by improved diagnostic tools and a better understanding of iron's role in these conditions. While the market presents substantial opportunities, certain restraints, such as the high cost of certain intravenous iron formulations and potential side effects, require careful consideration. Despite these challenges, the ongoing innovation by key players like Daiichi Sankyo, Vifor Pharma (CSL Limited), and Pfizer, coupled with strategic collaborations and market expansion initiatives, indicates a strong and sustained growth outlook for the Intravenous Iron Drugs market.
Intravenous Iron Drugs Industry Company Market Share

Intravenous Iron Drugs Industry: Market Size, Trends, and Forecast 2025-2033
This comprehensive report delves deep into the Intravenous Iron Drugs Industry, a critical segment of the global healthcare market. Analyze the market composition, intricate trends, and strategic forecast from 2019 to 2033, with a base year of 2025. Discover the market's evolution, propelled by groundbreaking product innovations, expanding applications, and shifting patient needs. This report offers unparalleled insights into the market dynamics, competitive landscape, and future trajectory of IV iron therapy, essential for stakeholders seeking to navigate and capitalize on this burgeoning sector.
Intravenous Iron Drugs Industry Market Composition & Trends
The Intravenous Iron Drugs Industry is characterized by a dynamic market composition, driven by intense competition and ongoing innovation. While specific market share distribution percentages are proprietary, the market is notably concentrated among a few key players, with a significant portion of the global revenue attributed to leading manufacturers. Innovation catalysts include the persistent need for effective anemia management, particularly in chronic conditions, and the development of novel drug delivery systems offering improved safety and efficacy profiles. The regulatory landscape plays a crucial role, with stringent approval processes by bodies like the FDA and EMA shaping market entry and product lifecycles. Substitute products, primarily oral iron supplements, remain a consideration, but their limitations in cases of malabsorption or rapid iron repletion often drive demand towards IV iron therapies. End-user profiles are diverse, encompassing patients with Chronic Kidney Disease (CKD), Irritable Bowel Disease (IBD), cancer-related anemia, and post-pregnancy anemia, among others. Merger and acquisition (M&A) activities are a strategic tool for market consolidation and expansion, with significant deal values often observed as companies seek to acquire complementary technologies or broaden their product portfolios. The overall market value is projected to reach tens of billions of dollars by 2025.
Intravenous Iron Drugs Industry Industry Evolution
The Intravenous Iron Drugs Industry has witnessed a remarkable evolution over the historical period of 2019-2024, and its growth trajectory is poised for significant acceleration through the forecast period of 2025-2033. The market's expansion is underpinned by a confluence of factors, including an increasing global prevalence of iron deficiency anemia (IDA), a growing understanding of its impact on various chronic diseases, and substantial advancements in pharmaceutical technology. During the historical period, market growth rates averaged approximately XX% annually, driven by the rising diagnosis of IDA in patient populations with Chronic Kidney Disease (CKD) and Cancer. Technological advancements have been pivotal, with the development of newer generations of intravenous iron formulations like Ferric Carboxymaltose and Iron Sucrose offering enhanced safety profiles and reduced infusion times compared to older Iron Dextran formulations. This has led to higher adoption metrics, with an estimated XX% of eligible patients now receiving IV iron therapy for severe anemia. Shifting consumer demands, or rather patient and physician preferences, have also influenced this evolution. There's a pronounced move towards therapies that offer convenience and minimize patient discomfort, pushing manufacturers to innovate in areas like patient-friendly dosing regimens and reduced hypersensitivity reactions. Furthermore, the increasing focus on patient-centric care and improved quality of life for individuals suffering from chronic conditions has amplified the demand for effective and rapid iron replenishment solutions. The market has transitioned from being a niche treatment for severe anemia to a more mainstream therapeutic option for a broader spectrum of iron deficiency conditions, reflecting a maturity in both product development and market penetration. The global market size, valued at billions in 2019, has steadily grown, with projections indicating continued double-digit growth in the coming years.
Leading Regions, Countries, or Segments in Intravenous Iron Drugs Industry
The dominance within the Intravenous Iron Drugs Industry is multifaceted, with certain regions and product segments exhibiting exceptional growth and market penetration. North America, particularly the United States, consistently leads the market, driven by a high prevalence of Chronic Kidney Disease (CKD) and a well-established healthcare infrastructure that facilitates rapid adoption of advanced therapies. The region's robust reimbursement policies and significant research and development investments further solidify its leading position. In terms of Product Type, Ferric Carboxymaltose has emerged as a frontrunner, showcasing superior efficacy and a favorable safety profile for a wide range of applications. Its market share is projected to expand considerably due to its versatility and physician preference.
Key drivers for regional and segmental dominance include:
- Investment Trends: Significant R&D investments in North America and Europe are fueling the development of next-generation IV iron therapies.
- Regulatory Support: Favorable regulatory pathways and accelerated approval processes for innovative IV iron drugs in key markets like the U.S. expedite market entry.
- Healthcare Infrastructure: Advanced healthcare systems in developed nations enable widespread access to IV iron infusion services.
- Prevalence of Chronic Diseases: The high incidence of CKD, IBD, and cancer in leading regions directly correlates with the demand for IV iron treatments.
Within the Application segment, Chronic Kidney Disease (CKD) remains the largest and most significant driver of the intravenous iron market. Patients with CKD often suffer from impaired erythropoiesis and iron malabsorption, necessitating efficient iron supplementation. The increasing global burden of CKD, coupled with a growing awareness of the benefits of IV iron in managing anemia in these patients, fuels substantial market demand. Following closely is the application in Cancer treatment, where chemotherapy-induced anemia and iron deficiency are common complications, making IV iron a vital supportive therapy. The market for Irritable Bowel Disease (IBD) applications is also experiencing steady growth as understanding of iron deficiency in these inflammatory conditions deepens.
Intravenous Iron Drugs Industry Product Innovations
Product innovation within the Intravenous Iron Drugs Industry is intensely focused on enhancing patient safety, optimizing therapeutic efficacy, and improving convenience. Breakthroughs have led to the development of complex iron carbohydrate nanoparticles, designed for targeted delivery and reduced immunogenicity, thereby minimizing adverse reactions like hypersensitivity. For instance, Ferric Carboxymaltose formulations have revolutionized treatment by allowing for higher single-dose administrations, significantly reducing the number of infusion visits required. Performance metrics highlight improved hemoglobin response rates and faster correction of iron deficiency compared to older agents. Unique selling propositions include faster infusion times, reduced risk of anaphylaxis, and a broader therapeutic window, making these advanced IV iron drugs the preferred choice for managing anemia across diverse patient populations.
Propelling Factors for Intravenous Iron Drugs Industry Growth
Several pivotal factors are propelling the growth of the Intravenous Iron Drugs Industry. A primary driver is the increasing global prevalence of iron deficiency anemia (IDA), particularly in conjunction with chronic diseases like Chronic Kidney Disease (CKD) and Cancer. Advancements in pharmaceutical technology have led to the development of safer and more effective intravenous iron formulations, such as Ferric Carboxymaltose and Iron Sucrose, offering improved patient outcomes and tolerability compared to older iron dextran products. Furthermore, evolving clinical guidelines and increased physician awareness regarding the benefits of early and aggressive iron repletion in managing anemia in these patient groups are significantly contributing to market expansion. Favorable reimbursement policies in key markets also play a crucial role, ensuring accessibility and affordability for a larger patient base.
Obstacles in the Intravenous Iron Drugs Industry Market
Despite its robust growth, the Intravenous Iron Drugs Industry faces several significant obstacles. Stringent regulatory hurdles and the complex approval processes for new IV iron formulations can lead to prolonged development timelines and substantial investment requirements. Supply chain disruptions, particularly for raw materials and specialized manufacturing components, can impact product availability and lead to price volatility. The high cost associated with intravenous iron therapies, when compared to oral iron supplements, can also be a barrier, especially in price-sensitive healthcare markets and for patients with limited insurance coverage. Moreover, the potential for adverse reactions, though minimized with newer formulations, still necessitates careful monitoring and can lead to physician hesitation in certain patient groups.
Future Opportunities in Intravenous Iron Drugs Industry
The Intravenous Iron Drugs Industry is ripe with future opportunities. The expanding global prevalence of iron deficiency, coupled with an aging population and increased diagnosis rates of chronic diseases, presents a substantial and growing patient pool. Emerging markets in Asia-Pacific and Latin America, with their rapidly developing healthcare infrastructures and increasing disposable incomes, offer significant untapped potential for market penetration. Technological advancements, such as the development of novel targeted drug delivery systems and sustained-release formulations, promise to further enhance safety and efficacy, opening new therapeutic avenues. Furthermore, exploring new applications beyond traditional uses, such as in pre-operative anemia management and post-partum recovery, could unlock substantial market growth.
Major Players in the Intravenous Iron Drugs Industry Ecosystem
- Daiichi Sankyo Company Ltd
- Americal Reagent
- Shield Therapeutics Plc
- Keryx Biopharmaceuticals Inc*List Not Exhaustive
- Pharmacosmos A/S
- AbbVie (Allergan)
- Rockwell Medical
- Zydus Group
- CSL Limited (Vifor Pharma Management Ltd )
- Covis Pharma GmbH (AMAG Pharmaceuticals Inc)
- Pfizer Inc
- Sanofi S A
Key Developments in Intravenous Iron Drugs Industry Industry
- November 2022: CSL Vifor and Fresenius Kabi received China's National Medical Products Administration (NMPA) approval for Ferinject, an intravenous iron therapy for the treatment of iron deficiency in adult patients for whom oral iron preparations are ineffective, oral iron preparations cannot be used, or for whom there is a clinical need to deliver iron rapidly. This development significantly expands access to advanced IV iron therapy in a major global market.
- November 2022: A few NIH-funded researchers began a study of intravenous iron drug treatment for post-pregnancy anemia patients. The study will enroll an estimated 5,000 women in Bangladesh, India, Pakistan, the Democratic Republic of Congo, Kenya, Zambia, and Guatemala. This initiative highlights the growing recognition of IV iron's potential in addressing postpartum anemia and its global health implications.
Strategic Intravenous Iron Drugs Industry Market Forecast
The strategic Intravenous Iron Drugs Industry market forecast predicts continued robust growth, driven by an increasing global burden of iron deficiency anemia and its strong association with chronic diseases like CKD and cancer. The ongoing development and adoption of safer and more efficacious IV iron formulations, such as Ferric Carboxymaltose, will be a key growth catalyst. Furthermore, the expanding healthcare infrastructure and rising disposable incomes in emerging economies present substantial opportunities for market penetration. Favorable clinical guidelines and increasing physician awareness are expected to further bolster demand, positioning the IV iron market for sustained double-digit annual growth through the forecast period, reaching tens of billions in market value by 2033.
Intravenous Iron Drugs Industry Segmentation
-
1. Product Type
- 1.1. Ferric Carboxymaltose
- 1.2. Iron Sucrose
- 1.3. Iron Dextran
- 1.4. Others
-
2. Application
- 2.1. Chronic Kidney Disease
- 2.2. Irriable Bowel Disease
- 2.3. Cancer
- 2.4. Others
Intravenous Iron Drugs Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
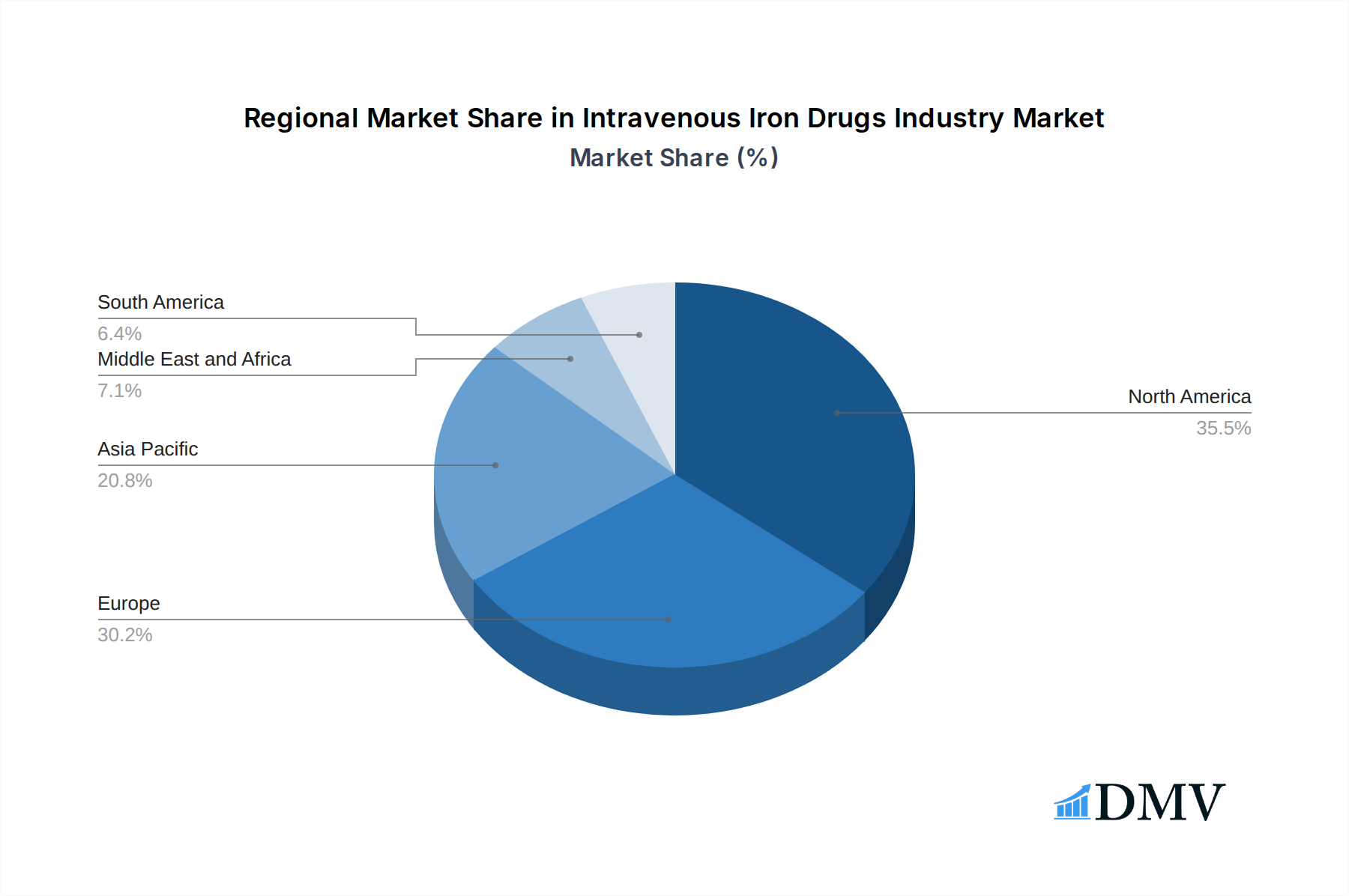
Intravenous Iron Drugs Industry Regional Market Share

Geographic Coverage of Intravenous Iron Drugs Industry
Intravenous Iron Drugs Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. DMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Ferric Carboxymaltose
- 5.1.2. Iron Sucrose
- 5.1.3. Iron Dextran
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Chronic Kidney Disease
- 5.2.2. Irriable Bowel Disease
- 5.2.3. Cancer
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East and Africa
- 5.3.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. Global Intravenous Iron Drugs Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Ferric Carboxymaltose
- 6.1.2. Iron Sucrose
- 6.1.3. Iron Dextran
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Chronic Kidney Disease
- 6.2.2. Irriable Bowel Disease
- 6.2.3. Cancer
- 6.2.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. North America Intravenous Iron Drugs Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 7.1.1. Ferric Carboxymaltose
- 7.1.2. Iron Sucrose
- 7.1.3. Iron Dextran
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Chronic Kidney Disease
- 7.2.2. Irriable Bowel Disease
- 7.2.3. Cancer
- 7.2.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 8. Europe Intravenous Iron Drugs Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 8.1.1. Ferric Carboxymaltose
- 8.1.2. Iron Sucrose
- 8.1.3. Iron Dextran
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Chronic Kidney Disease
- 8.2.2. Irriable Bowel Disease
- 8.2.3. Cancer
- 8.2.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 9. Asia Pacific Intravenous Iron Drugs Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 9.1.1. Ferric Carboxymaltose
- 9.1.2. Iron Sucrose
- 9.1.3. Iron Dextran
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Chronic Kidney Disease
- 9.2.2. Irriable Bowel Disease
- 9.2.3. Cancer
- 9.2.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 10. Middle East and Africa Intravenous Iron Drugs Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 10.1.1. Ferric Carboxymaltose
- 10.1.2. Iron Sucrose
- 10.1.3. Iron Dextran
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Chronic Kidney Disease
- 10.2.2. Irriable Bowel Disease
- 10.2.3. Cancer
- 10.2.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 11. South America Intravenous Iron Drugs Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 11.1.1. Ferric Carboxymaltose
- 11.1.2. Iron Sucrose
- 11.1.3. Iron Dextran
- 11.1.4. Others
- 11.2. Market Analysis, Insights and Forecast - by Application
- 11.2.1. Chronic Kidney Disease
- 11.2.2. Irriable Bowel Disease
- 11.2.3. Cancer
- 11.2.4. Others
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Daiichi Sankyo Company Ltd
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Americal Reagent
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Shield Therapeutics Plc
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Keryx Biopharmaceuticals Inc*List Not Exhaustive
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Pharmacosmos A/S
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 AbbVie (Allergan)
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Rockwell Medical
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Zydus Group
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 CSL Limited (Vifor Pharma Management Ltd )
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Covis Pharma GmbH (AMAG Pharmaceuticals Inc)
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Pfizer Inc
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Sanofi S A
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.1 Daiichi Sankyo Company Ltd
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Intravenous Iron Drugs Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Intravenous Iron Drugs Industry Revenue (billion), by Product Type 2025 & 2033
- Figure 3: North America Intravenous Iron Drugs Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 4: North America Intravenous Iron Drugs Industry Revenue (billion), by Application 2025 & 2033
- Figure 5: North America Intravenous Iron Drugs Industry Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Intravenous Iron Drugs Industry Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Intravenous Iron Drugs Industry Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe Intravenous Iron Drugs Industry Revenue (billion), by Product Type 2025 & 2033
- Figure 9: Europe Intravenous Iron Drugs Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 10: Europe Intravenous Iron Drugs Industry Revenue (billion), by Application 2025 & 2033
- Figure 11: Europe Intravenous Iron Drugs Industry Revenue Share (%), by Application 2025 & 2033
- Figure 12: Europe Intravenous Iron Drugs Industry Revenue (billion), by Country 2025 & 2033
- Figure 13: Europe Intravenous Iron Drugs Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Pacific Intravenous Iron Drugs Industry Revenue (billion), by Product Type 2025 & 2033
- Figure 15: Asia Pacific Intravenous Iron Drugs Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 16: Asia Pacific Intravenous Iron Drugs Industry Revenue (billion), by Application 2025 & 2033
- Figure 17: Asia Pacific Intravenous Iron Drugs Industry Revenue Share (%), by Application 2025 & 2033
- Figure 18: Asia Pacific Intravenous Iron Drugs Industry Revenue (billion), by Country 2025 & 2033
- Figure 19: Asia Pacific Intravenous Iron Drugs Industry Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East and Africa Intravenous Iron Drugs Industry Revenue (billion), by Product Type 2025 & 2033
- Figure 21: Middle East and Africa Intravenous Iron Drugs Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 22: Middle East and Africa Intravenous Iron Drugs Industry Revenue (billion), by Application 2025 & 2033
- Figure 23: Middle East and Africa Intravenous Iron Drugs Industry Revenue Share (%), by Application 2025 & 2033
- Figure 24: Middle East and Africa Intravenous Iron Drugs Industry Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East and Africa Intravenous Iron Drugs Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Intravenous Iron Drugs Industry Revenue (billion), by Product Type 2025 & 2033
- Figure 27: South America Intravenous Iron Drugs Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 28: South America Intravenous Iron Drugs Industry Revenue (billion), by Application 2025 & 2033
- Figure 29: South America Intravenous Iron Drugs Industry Revenue Share (%), by Application 2025 & 2033
- Figure 30: South America Intravenous Iron Drugs Industry Revenue (billion), by Country 2025 & 2033
- Figure 31: South America Intravenous Iron Drugs Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Product Type 2020 & 2033
- Table 2: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 3: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Product Type 2020 & 2033
- Table 5: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 6: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Product Type 2020 & 2033
- Table 11: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 12: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Germany Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United Kingdom Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: France Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Italy Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 17: Spain Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Rest of Europe Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 19: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Product Type 2020 & 2033
- Table 20: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 21: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 22: China Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Japan Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: India Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Australia Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: South Korea Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Asia Pacific Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Product Type 2020 & 2033
- Table 29: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 30: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 31: GCC Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: South Africa Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: Rest of Middle East and Africa Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Product Type 2020 & 2033
- Table 35: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 36: Global Intravenous Iron Drugs Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 37: Brazil Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: Argentina Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 39: Rest of South America Intravenous Iron Drugs Industry Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Intravenous Iron Drugs Industry?
The projected CAGR is approximately 9.6%.
2. Which companies are prominent players in the Intravenous Iron Drugs Industry?
Key companies in the market include Daiichi Sankyo Company Ltd, Americal Reagent, Shield Therapeutics Plc, Keryx Biopharmaceuticals Inc*List Not Exhaustive, Pharmacosmos A/S, AbbVie (Allergan), Rockwell Medical, Zydus Group, CSL Limited (Vifor Pharma Management Ltd ), Covis Pharma GmbH (AMAG Pharmaceuticals Inc), Pfizer Inc, Sanofi S A.
3. What are the main segments of the Intravenous Iron Drugs Industry?
The market segments include Product Type, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 3.6 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Target Diseases; Disadvantages of Oral Iron Drugs.
6. What are the notable trends driving market growth?
Ferric Carboxy Maltose Segment is Expected to Hold a Significant Market Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Adverse Reactions associated with Intravenous Iron Drugs.
8. Can you provide examples of recent developments in the market?
In November 2022, CSL Vifor and Fresenius Kabi received China's National Medical Products Administration (NMPA) approval for Ferinject, an intravenous iron therapy for the treatment of iron deficiency in adult patients for whom oral iron preparations are ineffective, oral iron preparations cannot be used or for whom there is a clinical need to deliver iron rapidly.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Intravenous Iron Drugs Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Intravenous Iron Drugs Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Intravenous Iron Drugs Industry?
To stay informed about further developments, trends, and reports in the Intravenous Iron Drugs Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


