Key Insights
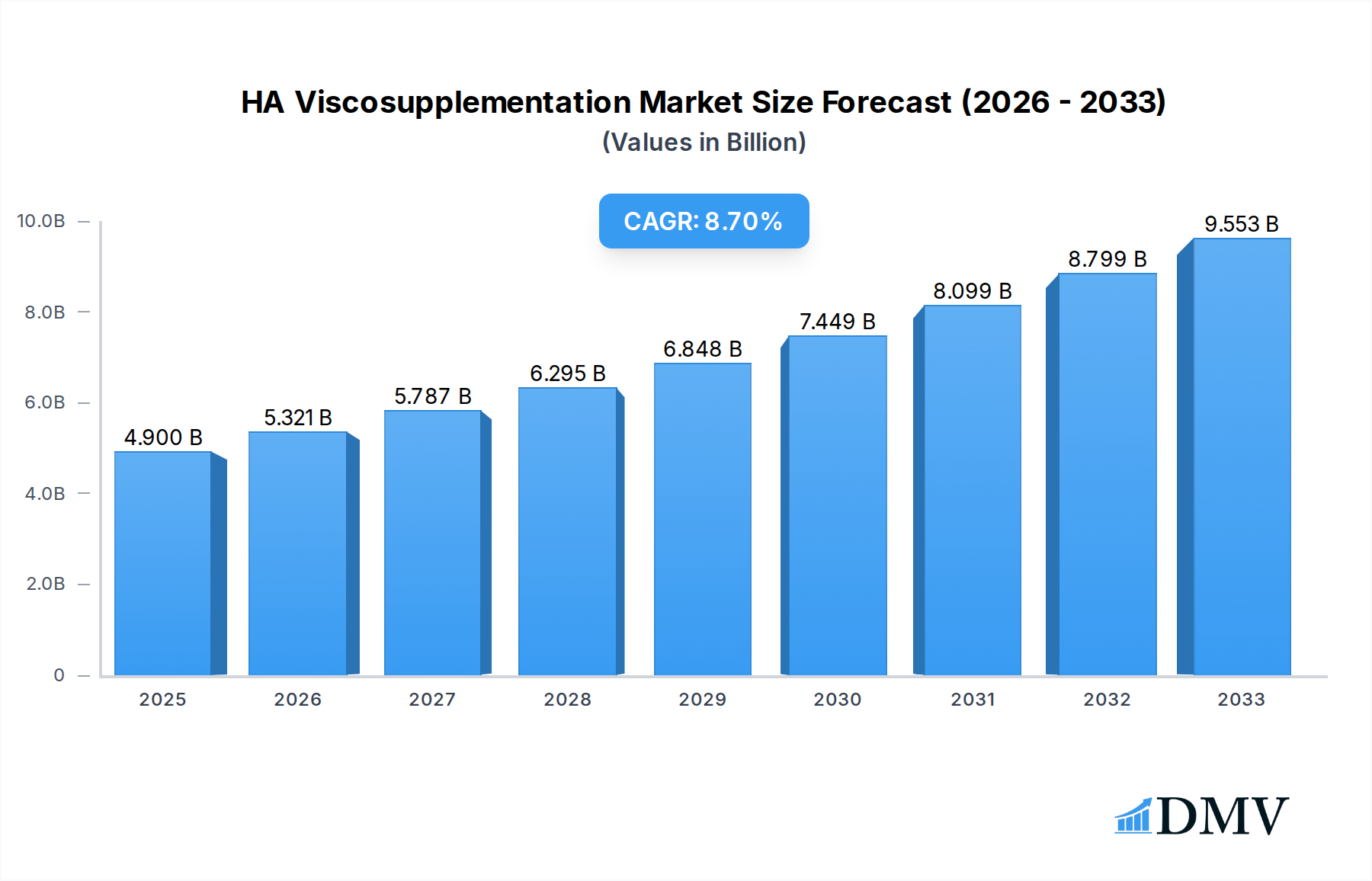
The global HA Viscosupplementation market is poised for significant expansion, projected to reach an estimated $4.9 billion in 2025, driven by an impressive 8.5% CAGR. This robust growth is primarily fueled by the increasing prevalence of osteoarthritis (OA), a degenerative joint disease that affects millions worldwide. As the global population ages, so does the incidence of OA, creating a sustained demand for effective pain management and joint function restoration solutions. Furthermore, advancements in HA viscosupplementation formulations, including longer-lasting injections and improved delivery systems, are enhancing treatment efficacy and patient satisfaction. The growing awareness among healthcare professionals and patients regarding the benefits of viscosupplementation, such as its minimally invasive nature and potential to delay or avoid surgery, is also a key growth accelerant. The market is segmented by application, with hospitals and clinics being the primary end-users due to their specialized infrastructure and patient volume. The types of injections, ranging from single to multiple injections, cater to diverse patient needs and treatment protocols.
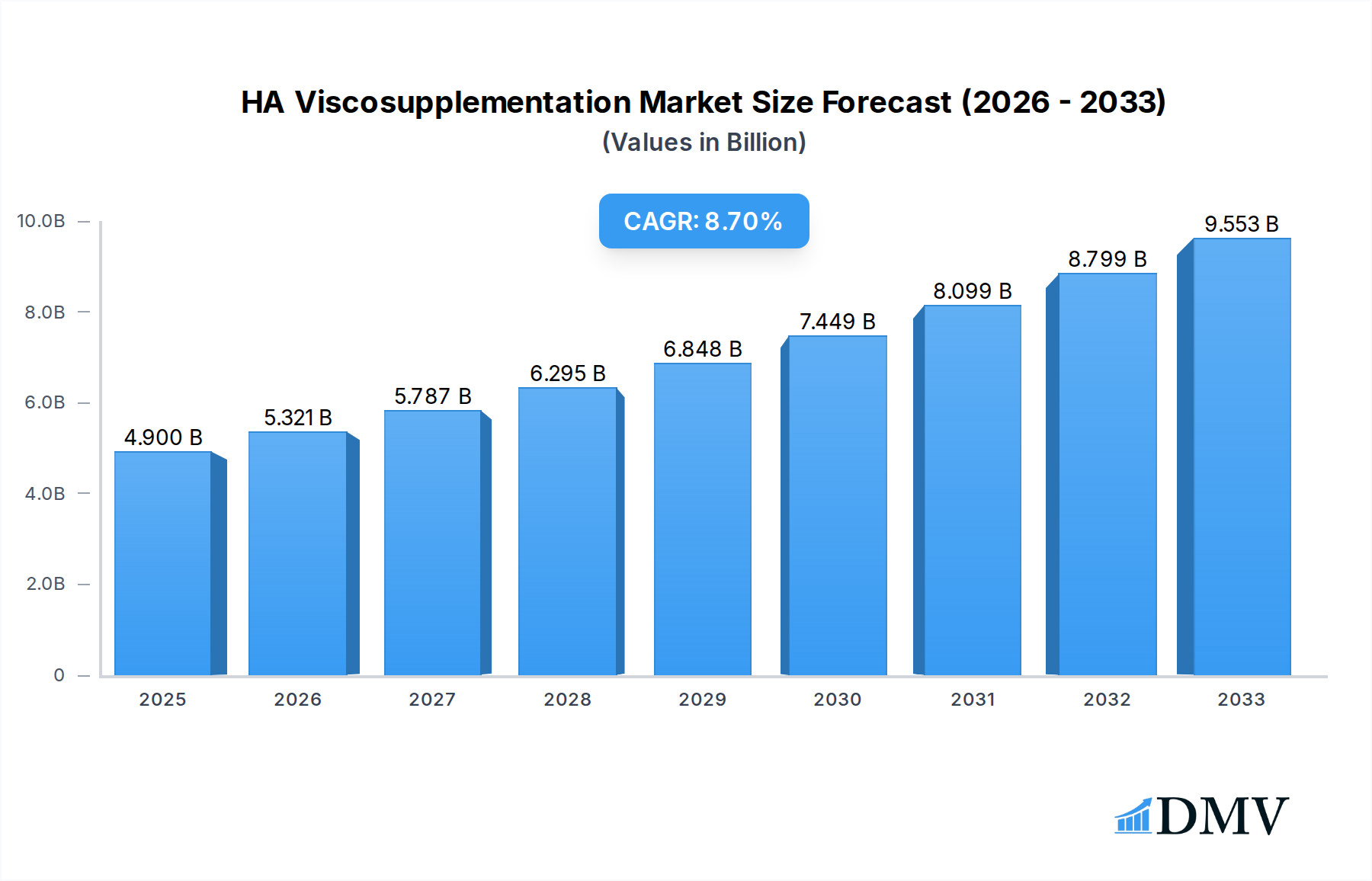
HA Viscosupplementation Market Size (In Billion)

Leading players such as Sanofi, Anika Therapeutics, and Zimmer Biomet are actively investing in research and development to innovate product pipelines and expand their market reach. This competitive landscape fosters the introduction of novel technologies and therapies, further stimulating market growth. Emerging economies, particularly in the Asia Pacific region, present significant untapped potential, driven by improving healthcare infrastructure, rising disposable incomes, and increasing access to advanced medical treatments. While the market enjoys strong growth drivers, potential restraints include the high cost of some advanced viscosupplementation treatments, which can limit accessibility for certain patient demographics, and the ongoing development and adoption of alternative OA treatment modalities like regenerative medicine and advanced surgical techniques. Nonetheless, the inherent benefits of HA viscosupplementation in managing OA symptoms and improving quality of life for patients are expected to sustain its upward trajectory in the foreseeable future.
HA Viscosupplementation Company Market Share

The HA Viscosupplementation market, a critical segment within the broader orthopedic solutions landscape, exhibits a dynamic composition influenced by robust innovation and evolving regulatory frameworks. Market concentration is moderate, with key players like Sanofi, Anika Therapeutics, and Seikagaku holding significant market shares, estimated to be in the hundreds of billions. The study period, from 2019 to 2033, with a base year of 2025, has witnessed substantial investment in R&D, driving a continuous stream of innovation catalysts. Regulatory bodies globally are actively shaping the market, with approvals for novel formulations and advanced delivery systems playing a crucial role. Substitute products, primarily oral pain medications and traditional surgical interventions, continue to pose a competitive threat, though the minimally invasive nature and efficacy of HA viscosupplementation offer distinct advantages. End-user profiles are expanding beyond traditional hospital settings to include specialized clinics and outpatient facilities, reflecting a shift towards decentralized care. Mergers and acquisitions (M&A) are a recurring theme, with deal values often reaching billions, signaling strategic consolidation and expansion of product portfolios. For instance, acquisitions by Zimmer Biomet and Bioventus have significantly altered market share distribution, projected to be in the hundreds of billions by 2025.
HA Viscosupplementation Industry Evolution
The HA Viscosupplementation industry has undergone a remarkable evolution, characterized by consistent market growth trajectories, driven by an increasing prevalence of osteoarthritis and a growing demand for minimally invasive treatment options. The study period (2019–2033), with a base year of 2025, highlights a CAGR of approximately XX% for the global market, which is projected to reach hundreds of billions by 2025. Technological advancements have been pivotal in this evolution, with the development of improved HA formulations offering enhanced viscoelastic properties, longer residence times, and better pain relief. These innovations, coupled with advancements in injection techniques, have led to higher patient satisfaction and improved clinical outcomes. Consumer demands are also shifting; patients are increasingly seeking treatments that allow for a faster return to daily activities and minimize downtime. This trend strongly favors viscosupplementation over more invasive procedures. The forecast period (2025–2033) anticipates continued expansion, fueled by an aging global population and rising healthcare expenditure in emerging economies. The historical period (2019–2024) laid the groundwork for this growth, with steady adoption rates and initial investments in product development. Companies such as LG Chem and Ferring have been at the forefront of these advancements, introducing next-generation products that cater to a wider range of patient needs. The market size is expected to cross hundreds of billions by the end of the forecast period.
Leading Regions, Countries, or Segments in HA Viscosupplementation
The HA Viscosupplementation market's dominance is intricately linked to specific regions, countries, and application segments, driven by a confluence of factors including robust healthcare infrastructure, favorable reimbursement policies, and high disease prevalence.
Dominant Application: Hospitals and Clinics
- High Treatment Volume: Hospitals and specialized orthopedic clinics represent the largest application segments for HA Viscosupplementation. This is due to their established infrastructure for performing orthopedic procedures and accommodating the growing number of osteoarthritis patients requiring treatment.
- Expertise and Resources: These healthcare settings possess the necessary medical expertise, advanced diagnostic tools, and therapeutic resources to effectively administer HA injections and manage patient care throughout the treatment journey.
- Reimbursement Landscape: Favorable reimbursement policies and insurance coverage for viscosupplementation procedures in many developed nations significantly drive adoption within these settings, contributing to an estimated market value in the hundreds of billions by 2025.
- Technological Integration: Hospitals and clinics are early adopters of new viscosupplementation technologies and product innovations, ensuring that patients have access to the latest and most effective treatments.
Dominant Type: Single Injection Formulations
- Patient Convenience and Compliance: Single-injection formulations are gaining significant traction due to their enhanced convenience for patients, requiring fewer clinic visits and reducing treatment burden. This leads to higher patient compliance and satisfaction.
- Cost-Effectiveness: For many patients and healthcare providers, single-injection treatments offer a more cost-effective solution compared to multi-injection regimens, contributing to their widespread adoption. The projected market share for single injections is expected to be in the hundreds of billions.
- Product Innovation Focus: Manufacturers are increasingly investing in the development of advanced single-injection products with improved efficacy and longer duration of action, further solidifying their market dominance.
- Growing Preference: The preference for single-injection therapies is projected to intensify throughout the forecast period (2025–2033), driven by ongoing technological advancements and a desire for streamlined treatment pathways.
Regional Dominance: North America and Europe
- Aging Population: The significant aging population in North America and Europe translates to a higher incidence of osteoarthritis, creating a substantial patient pool for viscosupplementation treatments.
- Advanced Healthcare Systems: These regions boast highly developed healthcare systems with extensive access to orthopedic specialists and advanced medical technologies, facilitating the widespread availability and adoption of HA viscosupplementation.
- Strong Research & Development: Significant investment in R&D by leading companies like Sanofi, Anika Therapeutics, and Zimmer Biomet in these regions drives product innovation and market growth, with market penetration reaching hundreds of billions.
- Reimbursement Policies: Supportive reimbursement frameworks and insurance coverage for viscosupplementation procedures in both North America and Europe are critical drivers of market expansion.
HA Viscosupplementation Product Innovations
Recent advancements in HA Viscosupplementation are revolutionizing patient outcomes and expanding treatment horizons. Innovations focus on enhanced rheological properties, leading to improved joint lubrication and shock absorption, with formulations demonstrating significantly longer residence times within the joint, often exceeding hundreds of days. Novel cross-linking technologies are yielding products with superior viscoelasticity and mechanical stability, providing sustained pain relief and functional improvement for osteoarthritis patients. Furthermore, research into biodegradable HA derivatives and the incorporation of therapeutic agents within HA matrices are paving the way for next-generation treatments. These product innovations are characterized by their minimally invasive nature, quick recovery times, and a strong emphasis on patient-specific treatment plans, significantly enhancing the overall value proposition and market appeal with an estimated market value in the hundreds of billions.
Propelling Factors for HA Viscosupplementation Growth
The HA Viscosupplementation market is propelled by a confluence of powerful factors. The escalating global prevalence of osteoarthritis, directly correlated with an aging demographic and lifestyle changes, creates a vast and growing patient pool. Technological advancements in HA formulation and delivery systems are consistently enhancing efficacy, safety, and patient convenience, making these treatments more attractive. Favorable reimbursement policies and increasing healthcare expenditure, particularly in emerging economies, are expanding access and affordability. Furthermore, the increasing preference for minimally invasive procedures over traditional surgery, driven by a desire for faster recovery and reduced complications, acts as a significant growth catalyst.
Obstacles in the HA Viscosupplementation Market
Despite its robust growth, the HA Viscosupplementation market faces several obstacles. Stringent regulatory approval processes for new formulations and devices can be time-consuming and costly, delaying market entry. The presence of established and effective alternative treatments, including oral pain relievers and physical therapy, presents a competitive challenge. Fluctuations in raw material prices and complexities in the supply chain can impact manufacturing costs and product availability. Furthermore, varying reimbursement rates across different regions and the need for continuous patient education to ensure proper understanding and compliance with treatment protocols can also act as barriers to wider adoption.
Future Opportunities in HA Viscosupplementation
The future of HA Viscosupplementation is rife with opportunities. The untapped potential in emerging markets, particularly in Asia-Pacific and Latin America, offers significant scope for market expansion driven by rising disposable incomes and increasing healthcare awareness. Continued research and development into combination therapies, integrating HA with regenerative medicine approaches like stem cell therapy or platelet-rich plasma (PRP), hold immense promise for enhanced joint repair and pain management. The development of novel drug delivery systems for HA, such as sustained-release formulations or implantable devices, could further improve treatment efficacy and patient adherence.
Major Players in the HA Viscosupplementation Ecosystem
- Sanofi
- Anika Therapeutics
- Seikagaku
- Zimmer Biomet
- Bioventus
- Ferring
- LG Chem
- Freda
- Haohai Biological
- Shanghai Jingfeng
Key Developments in HA Viscosupplementation Industry
- 2023 Q4: Launch of a novel multi-component HA formulation by Bioventus, promising enhanced viscoelasticity and extended pain relief, impacting hundreds of billions in market value.
- 2024 Q1: Sanofi announces positive Phase III trial results for a new generation HA viscosupplement, demonstrating significant improvements in joint function, projected to capture substantial market share.
- 2024 Q2: Anika Therapeutics acquires a key competitor, strengthening its portfolio of hyaluronic acid-based therapies and expanding its global footprint.
- 2024 Q3: Seikagaku Corporation receives regulatory approval for a novel single-injection HA product in Europe, catering to increasing patient demand for convenience.
- 2025: LG Chem and Ferring forge a strategic partnership to co-develop advanced HA viscosupplementation treatments, aiming to leverage their combined expertise.
- 2025: Zimmer Biomet introduces an innovative delivery device for HA injections, designed to improve precision and patient comfort, further solidifying its market position.
- 2026: Haohai Biological and Shanghai Jingfeng announce a joint venture to expand HA viscosupplementation manufacturing capabilities in Asia, addressing the region's growing demand.
- 2027: Freda launches a new bio-fermented HA product, emphasizing sustainability and cost-effectiveness in its production.
Strategic HA Viscosupplementation Market Forecast
The strategic HA Viscosupplementation market forecast indicates sustained and robust growth, driven by an aging global population and the relentless pursuit of effective, minimally invasive osteoarthritis treatments. Emerging opportunities in underserved markets, coupled with continued innovation in formulation and delivery technologies, will be pivotal. The growing integration of HA viscosupplementation with regenerative medicine techniques presents a significant avenue for future market expansion, promising enhanced therapeutic benefits and addressing unmet clinical needs. Continued investment in R&D and strategic collaborations among major players are expected to shape the market landscape, solidifying its position as a cornerstone of orthopedic pain management, with the market size projected to exceed hundreds of billions by 2033.
HA Viscosupplementation Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Others
-
2. Types
- 2.1. Single Injection
- 2.2. Three Injection
- 2.3. Five Injection
HA Viscosupplementation Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
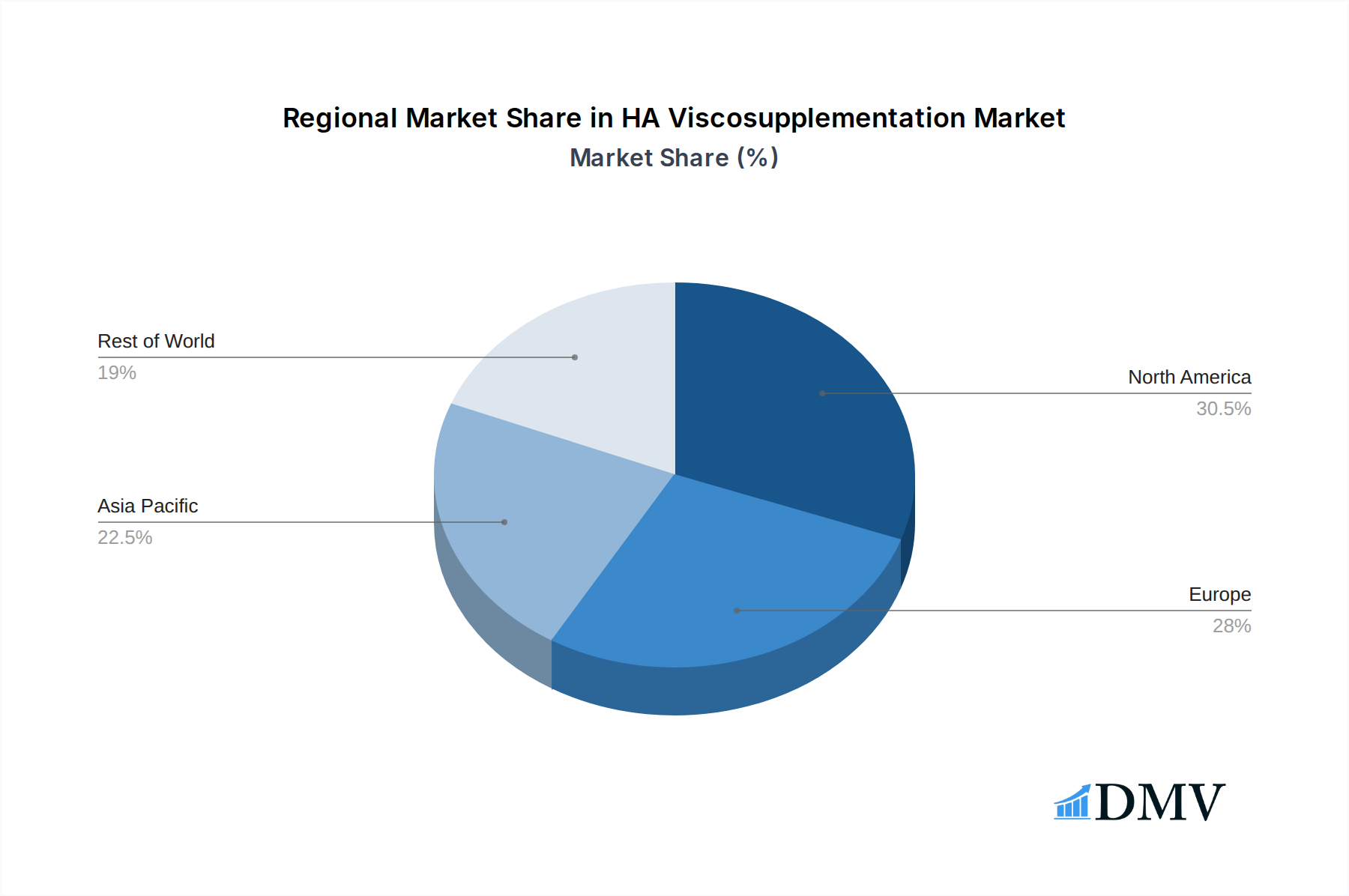
HA Viscosupplementation Regional Market Share

Geographic Coverage of HA Viscosupplementation
HA Viscosupplementation REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.5% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. DMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Single Injection
- 5.2.2. Three Injection
- 5.2.3. Five Injection
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global HA Viscosupplementation Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Single Injection
- 6.2.2. Three Injection
- 6.2.3. Five Injection
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America HA Viscosupplementation Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Single Injection
- 7.2.2. Three Injection
- 7.2.3. Five Injection
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America HA Viscosupplementation Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Single Injection
- 8.2.2. Three Injection
- 8.2.3. Five Injection
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe HA Viscosupplementation Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Single Injection
- 9.2.2. Three Injection
- 9.2.3. Five Injection
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa HA Viscosupplementation Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Single Injection
- 10.2.2. Three Injection
- 10.2.3. Five Injection
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific HA Viscosupplementation Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospitals
- 11.1.2. Clinics
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Single Injection
- 11.2.2. Three Injection
- 11.2.3. Five Injection
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Sanofi
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Anika Therapeutics
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Seikagaku
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Zimmer Biomet
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Bioventus
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Ferring
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 LG Chem
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Freda
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Haohai Biological
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Shanghai Jingfeng
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.1 Sanofi
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global HA Viscosupplementation Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America HA Viscosupplementation Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America HA Viscosupplementation Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America HA Viscosupplementation Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America HA Viscosupplementation Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America HA Viscosupplementation Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America HA Viscosupplementation Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America HA Viscosupplementation Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America HA Viscosupplementation Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America HA Viscosupplementation Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America HA Viscosupplementation Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America HA Viscosupplementation Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America HA Viscosupplementation Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe HA Viscosupplementation Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe HA Viscosupplementation Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe HA Viscosupplementation Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe HA Viscosupplementation Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe HA Viscosupplementation Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe HA Viscosupplementation Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa HA Viscosupplementation Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa HA Viscosupplementation Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa HA Viscosupplementation Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa HA Viscosupplementation Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa HA Viscosupplementation Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa HA Viscosupplementation Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific HA Viscosupplementation Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific HA Viscosupplementation Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific HA Viscosupplementation Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific HA Viscosupplementation Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific HA Viscosupplementation Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific HA Viscosupplementation Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global HA Viscosupplementation Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global HA Viscosupplementation Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global HA Viscosupplementation Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global HA Viscosupplementation Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global HA Viscosupplementation Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global HA Viscosupplementation Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global HA Viscosupplementation Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global HA Viscosupplementation Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global HA Viscosupplementation Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global HA Viscosupplementation Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global HA Viscosupplementation Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global HA Viscosupplementation Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global HA Viscosupplementation Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global HA Viscosupplementation Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global HA Viscosupplementation Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global HA Viscosupplementation Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global HA Viscosupplementation Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global HA Viscosupplementation Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific HA Viscosupplementation Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the HA Viscosupplementation?
The projected CAGR is approximately 8.5%.
2. Which companies are prominent players in the HA Viscosupplementation?
Key companies in the market include Sanofi, Anika Therapeutics, Seikagaku, Zimmer Biomet, Bioventus, Ferring, LG Chem, Freda, Haohai Biological, Shanghai Jingfeng.
3. What are the main segments of the HA Viscosupplementation?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 5600.00, USD 8400.00, and USD 11200.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "HA Viscosupplementation," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the HA Viscosupplementation report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the HA Viscosupplementation?
To stay informed about further developments, trends, and reports in the HA Viscosupplementation, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


