Key Insights
The China in-vitro diagnostics (IVD) market, valued at $10.61 billion in 2025, is poised for substantial growth, exhibiting a Compound Annual Growth Rate (CAGR) of 6.14% from 2025 to 2033. This expansion is driven by several key factors. Firstly, the rising prevalence of chronic diseases such as diabetes, cancer, and cardiovascular diseases in China fuels the demand for accurate and timely diagnostics. Secondly, increasing government initiatives to improve healthcare infrastructure and access, coupled with a growing focus on preventative healthcare, are significantly boosting the market. Technological advancements, particularly in molecular diagnostics and automation, are also contributing to market growth by enhancing diagnostic accuracy, speed, and efficiency. The market is segmented across various test types (clinical chemistry, molecular diagnostics, immunodiagnostics, hematology, and others), product categories (instruments, reagents, and other products), usability (disposable and reusable devices), applications (infectious diseases, diabetes, cancer, cardiology, and more), and end-users (diagnostic laboratories, hospitals, and clinics). The competitive landscape is robust, with both domestic and international players vying for market share. Major players include BioMerieux SA, Bio-Rad Laboratories Inc, BGI Group, QIAGEN NV, and Abbott Laboratories, among others. The continued expansion of the middle class and increased healthcare expenditure are expected to further propel market growth throughout the forecast period.
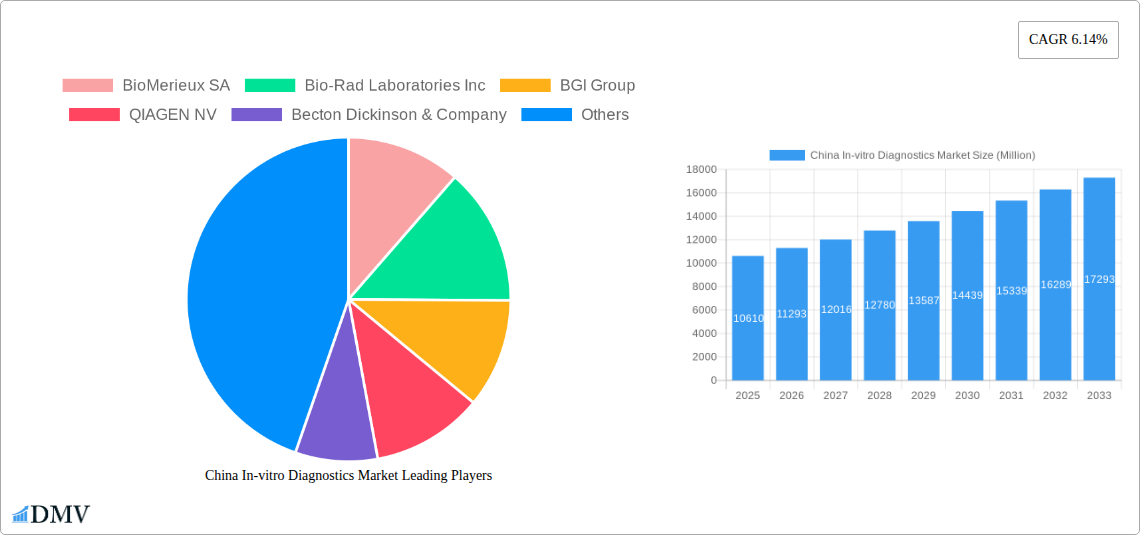
China In-vitro Diagnostics Market Market Size (In Billion)

The market's growth trajectory is expected to remain strong, fueled by continuous technological innovation and the expanding healthcare sector in China. However, factors like stringent regulatory requirements and pricing pressures could pose challenges. The increasing adoption of point-of-care diagnostics and telemedicine solutions is likely to reshape market dynamics in the coming years. The segments with the highest growth potential include molecular diagnostics, driven by advancements in genomics and personalized medicine, and point-of-care testing, addressing the need for rapid and accessible diagnostics in remote areas. Strategic collaborations and mergers and acquisitions are expected to be prominent strategies adopted by key players to consolidate their position and expand their product portfolios. The focus on developing cost-effective solutions while maintaining high quality is crucial for success in this competitive market.
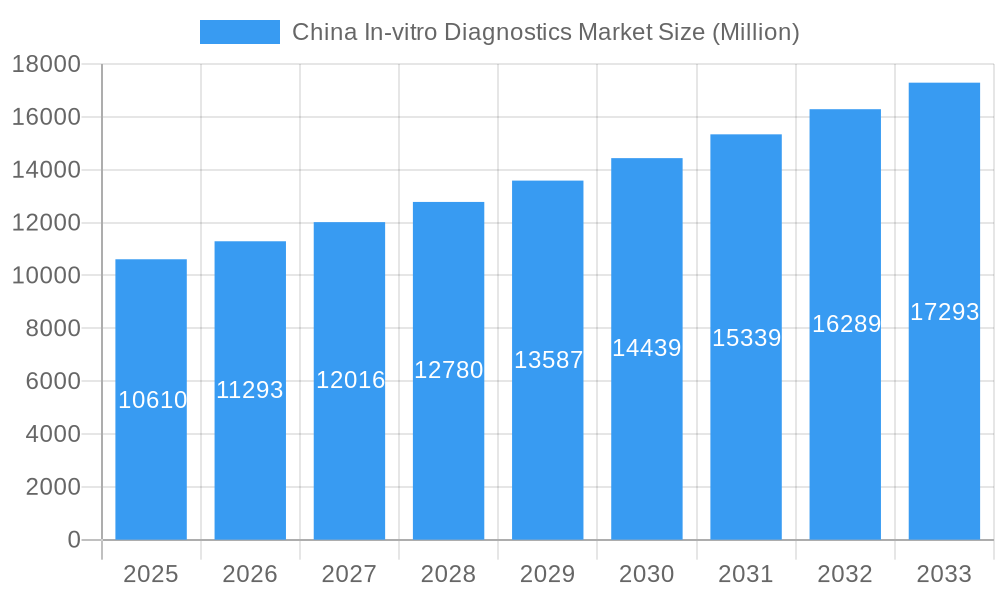
China In-vitro Diagnostics Market Company Market Share

China In-vitro Diagnostics Market: A Comprehensive Report (2019-2033)
This insightful report provides a detailed analysis of the burgeoning China in-vitro diagnostics (IVD) market, projecting robust growth from 2025 to 2033. It offers a comprehensive overview of market dynamics, competitive landscape, and future opportunities, equipping stakeholders with critical intelligence for strategic decision-making. The study period spans 2019-2033, with 2025 serving as the base and estimated year. The forecast period extends from 2025 to 2033, encompassing historical data from 2019-2024. The market is segmented by test type (Clinical Chemistry, Molecular Diagnostics, Immunodiagnostics, Hematology, Other Types), product (Instrument, Reagent, Other Products), usability (Disposable IVD Devices, Reusable IVD Devices), application (Infectious Disease, Diabetes, Cancer/Oncology, Cardiology, Autoimmune Disease, Nephrology, Other Applications), and end-user (Diagnostic Laboratories, Hospitals and Clinics, Other End-Users). Key players include BioMerieux SA, Bio-Rad Laboratories Inc, BGI Group, QIAGEN NV, Becton Dickinson & Company, Maccura Biotechnology Co Ltd, Abbott Laboratories, Shanghai Kehua Bio-Engineering Co Ltd, Danaher Corporation, Mindray Medical International Limited, F Hoffmann-La Roche AG, Autobio Diagnostics Co, Thermofisher Scientific Inc, Xiamen Boson Biotech Co Ltd, and Arkray Inc. The market size is projected to reach xx Million by 2033.
China In-vitro Diagnostics Market Composition & Trends
The China IVD market exhibits a dynamic interplay of factors shaping its evolution. Market concentration is moderately high, with several multinational corporations and established domestic players holding significant market share. However, the presence of numerous smaller, specialized companies fosters competition and innovation. The regulatory landscape, while evolving towards greater stringency, presents both challenges and opportunities. The substitution of older technologies by newer, more sophisticated techniques is a continuous process. The M&A landscape is active, with deal values ranging from xx Million to xx Million in recent years, reflecting strategic consolidation within the sector. End-user profiles encompass a diverse range of diagnostic laboratories, hospitals, and clinics, with diagnostic laboratories holding the largest share (xx%).
- Market Share Distribution: Top 5 players hold approximately xx% of the market.
- M&A Activity (2019-2024): xx deals totaling approximately xx Million USD in value.
- Regulatory Landscape: Emphasis on improving quality control and streamlining approval processes.
- Substitute Products: Continuous development and adoption of advanced diagnostic technologies impact market share.
China In-vitro Diagnostics Market Industry Evolution
The China IVD market has witnessed substantial and dynamic growth, characterized by a robust CAGR of **[Insert historical CAGR]%** during the historical period (2019-2024). This expansion has been significantly fueled by a confluence of factors, including escalating healthcare expenditure, a rising prevalence of chronic and infectious diseases, and proactive government initiatives aimed at bolstering healthcare infrastructure development and accessibility. Technological advancements have been a pivotal force, particularly in the realms of molecular diagnostics and automation, leading to marked improvements in diagnostic capabilities, accuracy, and overall efficiency. Consumer demand is increasingly gravitating towards diagnostic solutions that offer enhanced precision, faster turnaround times, and greater cost-effectiveness. The market is undergoing a significant transition, moving away from traditional diagnostic methodologies towards cutting-edge technologies such as Next-Generation Sequencing (NGS) and Point-of-Care Testing (POCT). Furthermore, the integration of Artificial Intelligence (AI) and Big Data analytics is playing an increasingly crucial role in augmenting diagnostic accuracy and accelerating the speed of analysis. Sustained growth is anticipated to continue at a projected CAGR of **[Insert forecast CAGR]%** during the forecast period (2025-2033). Specific indicators of this evolution include an estimated **[Insert % increase in molecular diagnostic tests]%** increase in the adoption of molecular diagnostic tests and a **[Insert % rise in automated analyzers]%** rise in the usage of automated analyzers, underscoring the shift towards more sophisticated diagnostic approaches.
Leading Regions, Countries, or Segments in China In-vitro Diagnostics Market
The IVD market in China is exhibiting a pronounced growth trajectory, with the advanced coastal regions, characterized by their well-established healthcare infrastructure and higher disposable incomes, leading the charge. Among the various market segments, Clinical Chemistry continues to command the largest market share, primarily due to the consistent high volume of routine diagnostic tests performed across healthcare settings. However, Molecular Diagnostics is emerging as a rapidly expanding segment, propelled by the escalating incidence of infectious diseases, the increasing awareness and diagnosis of genetic disorders, and the growing demand for personalized medicine. Within the product landscape, Reagents represent the largest market share, closely followed by diagnostic Instruments, reflecting the recurring need for consumable components. Disposable IVD devices are witnessing significant dominance due to their inherent convenience, ease of use, and crucial role in infection control protocols. Hospitals and clinical laboratories collectively constitute the major share of end-users, reflecting the centralized nature of diagnostic testing within the Chinese healthcare system.
- Key Drivers (Clinical Chemistry): Persistent high prevalence of chronic diseases such as diabetes, cardiovascular diseases, and cancer; well-established and extensive testing infrastructure in primary and secondary healthcare facilities; and continuous advancements in automation and reagent technology driving efficiency and throughput.
- Key Drivers (Molecular Diagnostics): The ongoing emergence and management of infectious diseases, including viral and bacterial infections; significant advancements and decreasing costs associated with Next-Generation Sequencing (NGS) technology, enabling broader genomic analysis; growing demand for genetic testing for disease predisposition and personalized treatment strategies; and increasing government support for infectious disease surveillance and research.
- Key Drivers (Hospitals and Clinics): Substantial government investment in modernizing healthcare infrastructure and expanding access to advanced medical services; an ever-increasing patient volume driven by an aging population and rising healthcare awareness; the growing adoption of integrated diagnostic platforms to improve workflow efficiency; and the demand for comprehensive diagnostic solutions to manage complex patient cases.
China In-vitro Diagnostics Market Product Innovations
Recent innovations focus on point-of-care diagnostics, offering faster and more convenient testing. Miniaturized instruments and integrated diagnostic platforms are gaining traction. Improvements in reagent technology enhance accuracy and sensitivity. The development of multiplexed assays enables simultaneous testing for multiple markers, improving diagnostic efficiency. Many innovations leverage AI to increase diagnostic accuracy and provide more detailed results. Several companies are developing rapid diagnostic tests for infectious diseases, emphasizing user-friendly designs and rapid turnaround times.
Propelling Factors for China In-vitro Diagnostics Market Growth
The robust growth of the China IVD market is propelled by a synergistic interplay of several key factors. Foremost among these are continuous technological advancements, including the development of highly sensitive rapid diagnostic tests for timely disease detection and the emergence of sophisticated AI-powered diagnostic tools that enhance analytical accuracy and predictive capabilities. Complementing this is the sustained economic growth and the subsequent rise in per capita healthcare spending, which directly translates into increased demand for advanced diagnostic services. Favorable government policies and strategic investments aimed at strengthening healthcare infrastructure, promoting the adoption of innovative medical technologies, and expanding health insurance coverage further serve as significant catalysts for market expansion. Additionally, the persistent and increasing prevalence of chronic diseases, such as cardiovascular conditions, diabetes, and various cancers, necessitates the widespread use of advanced and precise diagnostic tools, thereby driving market demand.
Obstacles in the China In-vitro Diagnostics Market
Despite its impressive growth, the China IVD market is not without its challenges. Stricter and evolving regulatory requirements for the approval of new diagnostic products can introduce significant delays in market entry, impacting the pace of innovation and commercialization. Potential supply chain disruptions, influenced by global economic factors, trade policies, or unforeseen events, can lead to shortages of critical reagents and instruments, causing price volatility and affecting accessibility. The market is also characterized by intense competition from both established domestic manufacturers and global IVD players, which can exert downward pressure on profit margins and necessitate continuous investment in research and development to maintain a competitive edge. These obstacles, if not adequately addressed, have the potential to moderate market growth and pose challenges to the seamless integration of groundbreaking diagnostic technologies.
Future Opportunities in China In-vitro Diagnostics Market
The future holds significant opportunities. Expansion into underserved rural areas promises growth. The development and adoption of personalized medicine approaches presents significant market potential. Technological advancements, such as improved POCT devices and AI-powered diagnostic platforms, will create new market niches. Increased focus on preventive healthcare initiatives will drive demand for diagnostic tools.
Major Players in the China In-vitro Diagnostics Market Ecosystem
- BioMerieux SA (BioMerieux SA)
- Bio-Rad Laboratories Inc (Bio-Rad Laboratories Inc)
- BGI Group (BGI Group)
- QIAGEN NV (QIAGEN NV)
- Becton Dickinson & Company (Becton Dickinson & Company)
- Maccura Biotechnology Co Ltd
- Abbott Laboratories (Abbott Laboratories)
- Shanghai Kehua Bio-Engineering Co Ltd
- Danaher Corporation (Danaher Corporation)
- Mindray Medical International Limited (Mindray Medical International Limited)
- F Hoffmann-La Roche AG (F Hoffmann-La Roche AG)
- Autobio Diagnostics Co
- Thermofisher Scientific Inc (Thermofisher Scientific Inc)
- Xiamen Boson Biotech Co Ltd
- Arkray Inc
Key Developments in China In-vitro Diagnostics Market Industry
- September 2023: Roche Diagnostics China Technical Innovation Center (TIC) opened in Shanghai, focusing on smart diagnostics and service innovation.
- March 2023: Hainan government issued new guidelines for administering urgently needed imported drugs and medical devices in the Boao Lecheng zone.
Strategic China In-vitro Diagnostics Market Forecast
The China IVD market is poised for continued expansion, driven by technological innovation, increasing healthcare expenditure, and supportive government policies. Emerging opportunities in personalized medicine and advanced diagnostic technologies present significant growth potential. The market's future trajectory indicates substantial growth, solidifying its position as a leading IVD market globally.
China In-vitro Diagnostics Market Segmentation
-
1. Test Type
- 1.1. Clinical Chemistry
- 1.2. Molecular Diagnostics
- 1.3. Immunodiagnostics
- 1.4. Hematology
- 1.5. Other Types
-
2. Product
- 2.1. Instrument
- 2.2. Reagent
- 2.3. Other Products
-
3. Usability
- 3.1. Disposable IVD Devices
- 3.2. Reusable IVD Devices
-
4. Application
- 4.1. Infectious Disease
- 4.2. Diabetes
- 4.3. Cancer/Oncology
- 4.4. Cardiology
- 4.5. Autoimmune Disease
- 4.6. Nephrology
- 4.7. Other Applications
-
5. End-User
- 5.1. Diagnostic Laboratories
- 5.2. Hospitals and Clinics
- 5.3. Other End-Users
China In-vitro Diagnostics Market Segmentation By Geography
- 1. China

China In-vitro Diagnostics Market Regional Market Share

Geographic Coverage of China In-vitro Diagnostics Market
China In-vitro Diagnostics Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.14% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.2.1. High Burden of Chronic Diseases and Infectious Diseases; Increasing Use of Point-of-Care (POC) Diagnostics Spurring the IVD Market; Increasing Awareness and Acceptance of Personalized Medicine
- 3.3. Market Restrains
- 3.3.1. Lack of Proper Reimbursement; Stringent Regulatory Framework
- 3.4. Market Trends
- 3.4.1. Molecular Diagnostics Segment Dominates the Chinese In Vitro Diagnostics Market
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. China In-vitro Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Test Type
- 5.1.1. Clinical Chemistry
- 5.1.2. Molecular Diagnostics
- 5.1.3. Immunodiagnostics
- 5.1.4. Hematology
- 5.1.5. Other Types
- 5.2. Market Analysis, Insights and Forecast - by Product
- 5.2.1. Instrument
- 5.2.2. Reagent
- 5.2.3. Other Products
- 5.3. Market Analysis, Insights and Forecast - by Usability
- 5.3.1. Disposable IVD Devices
- 5.3.2. Reusable IVD Devices
- 5.4. Market Analysis, Insights and Forecast - by Application
- 5.4.1. Infectious Disease
- 5.4.2. Diabetes
- 5.4.3. Cancer/Oncology
- 5.4.4. Cardiology
- 5.4.5. Autoimmune Disease
- 5.4.6. Nephrology
- 5.4.7. Other Applications
- 5.5. Market Analysis, Insights and Forecast - by End-User
- 5.5.1. Diagnostic Laboratories
- 5.5.2. Hospitals and Clinics
- 5.5.3. Other End-Users
- 5.6. Market Analysis, Insights and Forecast - by Region
- 5.6.1. China
- 5.1. Market Analysis, Insights and Forecast - by Test Type
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 BioMerieux SA
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Bio-Rad Laboratories Inc
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 BGI Group
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 QIAGEN NV
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 Becton Dickinson & Company
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Maccura Biotechnology Co Ltd
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Abbott Laboratories
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 Shanghai Kehua Bio-Engineering Co Ltd
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Danaher Corporation
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Mindray Medical International Limited
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 F Hoffmann-La Roche AG
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 Autobio Diagnostics Co
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.13 Thermofisher Scientific Inc
- 6.2.13.1. Overview
- 6.2.13.2. Products
- 6.2.13.3. SWOT Analysis
- 6.2.13.4. Recent Developments
- 6.2.13.5. Financials (Based on Availability)
- 6.2.14 Xiamen Boson Biotech Co Ltd
- 6.2.14.1. Overview
- 6.2.14.2. Products
- 6.2.14.3. SWOT Analysis
- 6.2.14.4. Recent Developments
- 6.2.14.5. Financials (Based on Availability)
- 6.2.15 Arkray Inc
- 6.2.15.1. Overview
- 6.2.15.2. Products
- 6.2.15.3. SWOT Analysis
- 6.2.15.4. Recent Developments
- 6.2.15.5. Financials (Based on Availability)
- 6.2.1 BioMerieux SA
List of Figures
- Figure 1: China In-vitro Diagnostics Market Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: China In-vitro Diagnostics Market Share (%) by Company 2025
List of Tables
- Table 1: China In-vitro Diagnostics Market Revenue Million Forecast, by Test Type 2020 & 2033
- Table 2: China In-vitro Diagnostics Market Volume K unit Forecast, by Test Type 2020 & 2033
- Table 3: China In-vitro Diagnostics Market Revenue Million Forecast, by Product 2020 & 2033
- Table 4: China In-vitro Diagnostics Market Volume K unit Forecast, by Product 2020 & 2033
- Table 5: China In-vitro Diagnostics Market Revenue Million Forecast, by Usability 2020 & 2033
- Table 6: China In-vitro Diagnostics Market Volume K unit Forecast, by Usability 2020 & 2033
- Table 7: China In-vitro Diagnostics Market Revenue Million Forecast, by Application 2020 & 2033
- Table 8: China In-vitro Diagnostics Market Volume K unit Forecast, by Application 2020 & 2033
- Table 9: China In-vitro Diagnostics Market Revenue Million Forecast, by End-User 2020 & 2033
- Table 10: China In-vitro Diagnostics Market Volume K unit Forecast, by End-User 2020 & 2033
- Table 11: China In-vitro Diagnostics Market Revenue Million Forecast, by Region 2020 & 2033
- Table 12: China In-vitro Diagnostics Market Volume K unit Forecast, by Region 2020 & 2033
- Table 13: China In-vitro Diagnostics Market Revenue Million Forecast, by Test Type 2020 & 2033
- Table 14: China In-vitro Diagnostics Market Volume K unit Forecast, by Test Type 2020 & 2033
- Table 15: China In-vitro Diagnostics Market Revenue Million Forecast, by Product 2020 & 2033
- Table 16: China In-vitro Diagnostics Market Volume K unit Forecast, by Product 2020 & 2033
- Table 17: China In-vitro Diagnostics Market Revenue Million Forecast, by Usability 2020 & 2033
- Table 18: China In-vitro Diagnostics Market Volume K unit Forecast, by Usability 2020 & 2033
- Table 19: China In-vitro Diagnostics Market Revenue Million Forecast, by Application 2020 & 2033
- Table 20: China In-vitro Diagnostics Market Volume K unit Forecast, by Application 2020 & 2033
- Table 21: China In-vitro Diagnostics Market Revenue Million Forecast, by End-User 2020 & 2033
- Table 22: China In-vitro Diagnostics Market Volume K unit Forecast, by End-User 2020 & 2033
- Table 23: China In-vitro Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 24: China In-vitro Diagnostics Market Volume K unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the China In-vitro Diagnostics Market?
The projected CAGR is approximately 6.14%.
2. Which companies are prominent players in the China In-vitro Diagnostics Market?
Key companies in the market include BioMerieux SA, Bio-Rad Laboratories Inc, BGI Group, QIAGEN NV, Becton Dickinson & Company, Maccura Biotechnology Co Ltd, Abbott Laboratories, Shanghai Kehua Bio-Engineering Co Ltd, Danaher Corporation, Mindray Medical International Limited, F Hoffmann-La Roche AG, Autobio Diagnostics Co, Thermofisher Scientific Inc, Xiamen Boson Biotech Co Ltd, Arkray Inc.
3. What are the main segments of the China In-vitro Diagnostics Market?
The market segments include Test Type, Product, Usability, Application, End-User.
4. Can you provide details about the market size?
The market size is estimated to be USD 10.61 Million as of 2022.
5. What are some drivers contributing to market growth?
High Burden of Chronic Diseases and Infectious Diseases; Increasing Use of Point-of-Care (POC) Diagnostics Spurring the IVD Market; Increasing Awareness and Acceptance of Personalized Medicine.
6. What are the notable trends driving market growth?
Molecular Diagnostics Segment Dominates the Chinese In Vitro Diagnostics Market.
7. Are there any restraints impacting market growth?
Lack of Proper Reimbursement; Stringent Regulatory Framework.
8. Can you provide examples of recent developments in the market?
September 2023, Roche Diagnostics China Technical Innovation Center (TIC) was opened in Shanghai's Pudong New Area in Jinqiao. It consists of laboratories and classrooms, covering 7,080 square meters focusing on a comprehensive upgrade of smart diagnostics, service innovation, talent training, and customer experience in diagnostics.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "China In-vitro Diagnostics Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the China In-vitro Diagnostics Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the China In-vitro Diagnostics Market?
To stay informed about further developments, trends, and reports in the China In-vitro Diagnostics Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


