Key Insights
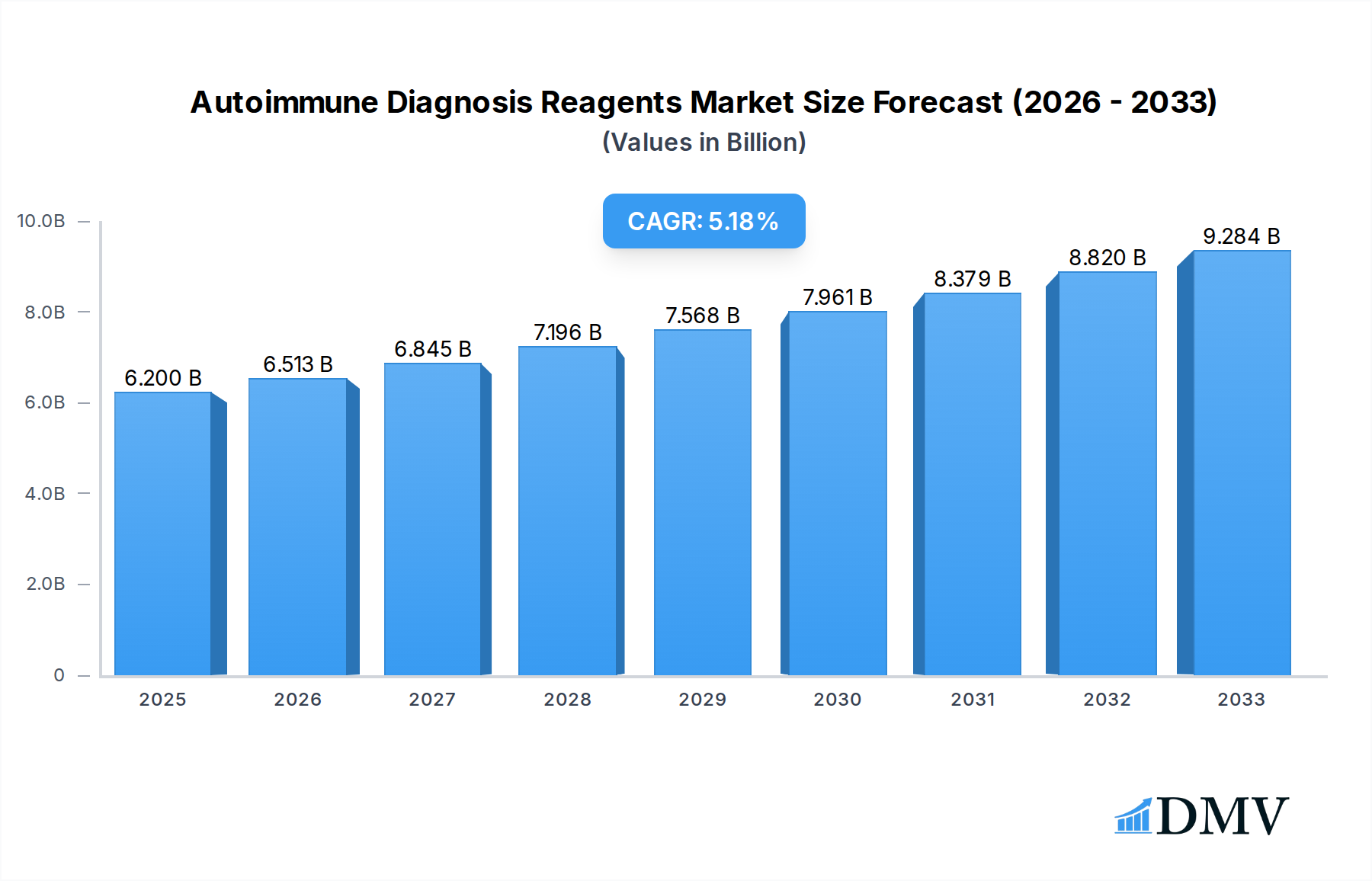
The global autoimmune diagnosis reagents market is poised for significant expansion, projected to reach an estimated USD 6.2 billion in 2025, driven by a robust compound annual growth rate (CAGR) of 5.1% through 2033. This growth is underpinned by a confluence of factors, including the increasing prevalence of autoimmune diseases worldwide, advancements in diagnostic technologies, and a growing awareness among healthcare professionals and patients regarding early and accurate diagnosis. The rising burden of conditions such as rheumatoid arthritis, lupus, and inflammatory bowel disease necessitates more sophisticated and reliable diagnostic tools, directly fueling demand for specialized reagents. Furthermore, the expanding healthcare infrastructure, particularly in emerging economies, and increased government initiatives to improve public health outcomes are contributing to market dynamism. The integration of automation and high-throughput screening in laboratories is also enhancing the efficiency and accuracy of autoimmune disease detection, further stimulating market penetration.
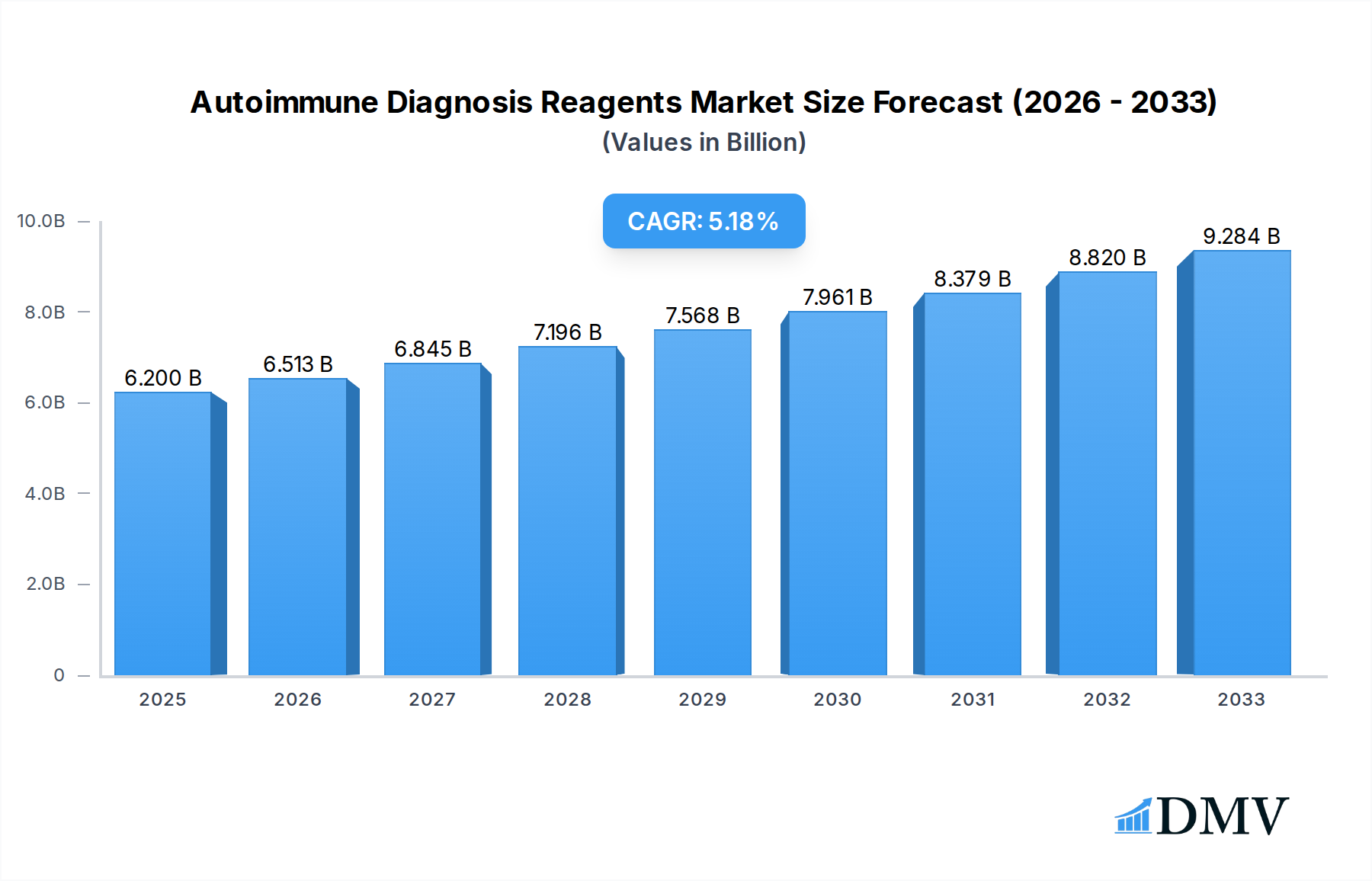
Autoimmune Diagnosis Reagents Market Size (In Billion)

The market segmentation reveals a diverse landscape, with applications spanning critical healthcare settings like hospitals and third-party testing agencies. In terms of disease types, rheumatology, hepatology, gastroenterology, and endocrinology represent key areas where these diagnostic reagents find extensive application. The market is characterized by the presence of prominent global players such as Thermo Scientific Phadia, Siemens Healthineers, and EUROIMMUN, alongside emerging innovators. Geographic analysis indicates that North America and Europe currently dominate the market due to advanced healthcare systems and high research & development expenditure. However, the Asia Pacific region is expected to witness the fastest growth, propelled by a burgeoning patient population, improving healthcare access, and increasing investments in diagnostic infrastructure. Restraints, such as the high cost of advanced diagnostic platforms and stringent regulatory approvals for new reagents, are being navigated through continuous innovation and strategic collaborations aimed at improving accessibility and affordability.
Autoimmune Diagnosis Reagents Company Market Share

Autoimmune Diagnosis Reagents Market Composition & Trends
The global Autoimmune Diagnosis Reagents market is characterized by a dynamic landscape with significant market concentration among leading players, driven by continuous innovation and a complex regulatory environment. The market share distribution shows dominant positions held by companies such as Thermo Scientific Phadia and Siemens Healthineers, collectively accounting for an estimated 25% of the market by revenue in the base year 2025, with a projected increase to XXX billion. EUROIMMUN and Dr. Fooke Laboratorien are also substantial contributors, each holding approximately XXX billion in market share. The innovation catalysts are primarily focused on improving assay sensitivity, specificity, and automation, catering to the increasing demand for early and accurate diagnosis of autoimmune diseases. Regulatory landscapes, while providing a framework for product approval and quality control, also present stringent requirements that impact market entry and product development timelines. Substitute products, such as advanced imaging techniques and genetic testing, are emerging but currently supplement rather than replace the core diagnostic reagent market. End-user profiles are diverse, encompassing hospitals, third-party testing agencies, and specialized diagnostic laboratories, each with distinct purchasing behaviors and technical requirements. Mergers and acquisitions (M&A) activities are on the rise, with significant deal values in the range of XXX billion to XXX billion in the historical period 2019-2024, signaling consolidation and strategic expansion by key players to enhance their product portfolios and geographic reach. For instance, a notable M&A in 2023 involved a deal valued at XXX billion.
- Market Share Distribution (Base Year 2025):
- Thermo Scientific Phadia: XXX billion
- Siemens Healthineers: XXX billion
- EUROIMMUN: XXX billion
- Dr. Fooke Laboratorien: XXX billion
- Others: XXX billion
- M&A Deal Values (Historical Period 2019-2024):
- Average Deal Value: XXX billion
- Total M&A Value: XXX billion
- Innovation Focus: Enhanced assay sensitivity, specificity, automation, point-of-care diagnostics.
- End-User Segments: Hospitals, third-party testing agencies, research institutions.
Autoimmune Diagnosis Reagents Industry Evolution
The Autoimmune Diagnosis Reagents industry has undergone a remarkable evolution, transforming from niche diagnostic tools to indispensable components of modern healthcare. Over the study period of 2019–2033, the market has witnessed a consistent growth trajectory, propelled by a confluence of factors including increasing prevalence of autoimmune diseases, advancements in diagnostic technologies, and a growing awareness among healthcare professionals and patients. The historical period from 2019 to 2024 saw the market expand from approximately XXX billion to an estimated XXX billion, demonstrating a compound annual growth rate (CAGR) of XXX%. This growth was underpinned by a heightened understanding of the complex mechanisms of autoimmune disorders and the critical need for early and precise diagnostic methods. Technological advancements have been at the forefront of this evolution, with significant investments directed towards developing highly sensitive and specific immunoassay platforms, including enzyme-linked immunosorbent assays (ELISA), chemiluminescence immunoassays (CLIA), and multiplex assays. These innovations have enabled the simultaneous detection of multiple autoantibodies, streamlining diagnostic workflows and improving diagnostic accuracy for conditions spanning Rheumatology, Hepatology, Gastroenterology, and Endocrinology.
Shifting consumer demands, largely driven by a proactive approach to healthcare and the desire for personalized medicine, have also played a pivotal role. Patients and clinicians alike are increasingly seeking diagnostic solutions that offer rapid turnaround times, minimal invasiveness, and comprehensive information. This has fueled the demand for automated systems and point-of-care testing (POCT) solutions within the autoimmune diagnostics space, leading to the development of benchtop analyzers and integrated platforms that can be deployed in diverse healthcare settings, from large hospitals to smaller clinics. The introduction of novel biomarkers and the refinement of existing antibody panels have further broadened the diagnostic capabilities of these reagents, allowing for earlier detection and better disease management. The market's robust growth is further evidenced by an estimated market size of XXX billion in the base year 2025, projected to reach a substantial XXX billion by the end of the forecast period in 2033, with a CAGR of XXX% from 2025 to 2033. This sustained expansion underscores the enduring importance and the promising future of autoimmune diagnosis reagents in addressing a significant global health challenge.
Leading Regions, Countries, or Segments in Autoimmune Diagnosis Reagents
The global Autoimmune Diagnosis Reagents market is led by North America, particularly the United States, due to its advanced healthcare infrastructure, high prevalence of autoimmune diseases, and significant R&D investments. The region's dominance is further amplified by a robust presence of key market players and a strong emphasis on early disease detection and management. In the United States, the Application segment of Hospitals is the largest contributor, accounting for an estimated XXX billion in revenue in 2025. This is attributed to the high volume of diagnostic tests performed in hospital settings and the integrated nature of healthcare delivery, facilitating comprehensive autoimmune disease workups. Third-party testing agencies also represent a significant segment, with a projected market size of XXX billion in 2025, driven by their specialized expertise and cost-effectiveness for certain diagnostic panels.
Within the Types segment, Rheumatology remains the largest and most influential category. In 2025, the Rheumatology segment is estimated to contribute XXX billion to the market. This dominance stems from the high incidence of rheumatological autoimmune diseases such as rheumatoid arthritis, lupus erythematosus, and Sjögren's syndrome, which necessitate extensive antibody profiling. The continuous development of new autoantibody targets and improved assay sensitivity for these conditions further solidifies Rheumatology's leading position. Hepatology and Gastroenterology are also substantial segments, with projected market values of XXX billion and XXX billion respectively in 2025, reflecting the increasing diagnosis of autoimmune liver diseases and inflammatory bowel diseases. Endocrinology follows closely, with the market valued at XXX billion in 2025, driven by the rise in autoimmune endocrine disorders like Type 1 diabetes and autoimmune thyroid diseases. The "Others" category, encompassing less prevalent autoimmune conditions and emerging research areas, is also witnessing steady growth, projected at XXX billion in 2025.
- Dominant Region: North America (United States)
- Key Drivers: Advanced healthcare infrastructure, high disease prevalence, strong R&D focus, presence of leading manufacturers.
- Dominant Application Segment: Hospitals
- Projected Market Size (2025): XXX billion
- Contributing Factors: High diagnostic test volumes, integrated healthcare systems, demand for comprehensive disease management.
- Dominant Type Segment: Rheumatology
- Projected Market Size (2025): XXX billion
- Driving Factors: High incidence of rheumatological autoimmune diseases, extensive antibody profiling needs, continuous development of novel biomarkers and assays.
- Other Significant Segments:
- Third-party Testing Agencies: XXX billion (2025)
- Hepatology: XXX billion (2025)
- Gastroenterology: XXX billion (2025)
- Endocrinology: XXX billion (2025)
- Others: XXX billion (2025)
Autoimmune Diagnosis Reagents Product Innovations
The Autoimmune Diagnosis Reagents market is continuously propelled by groundbreaking product innovations aimed at enhancing diagnostic accuracy, speed, and workflow efficiency. Recent advancements include the development of highly multiplexed assays capable of detecting dozens of autoantibodies simultaneously from a single small sample, significantly reducing hands-on time and improving the diagnostic yield. Novel assay formats leveraging chemiluminescent and fluorescent technologies offer superior sensitivity and dynamic range, enabling the detection of even low-affinity autoantibodies crucial for early diagnosis. Furthermore, the integration of sophisticated algorithms and AI-driven interpretation tools is revolutionizing how diagnostic data is analyzed, leading to more precise and clinically relevant results. The focus is shifting towards automated, fully integrated systems that minimize human error and expedite the reporting of results, making these advanced diagnostics more accessible across various healthcare settings.
Propelling Factors for Autoimmune Diagnosis Reagents Growth
The projected growth of the Autoimmune Diagnosis Reagents market is fueled by several key factors. The increasing incidence and prevalence of autoimmune diseases worldwide, driven by genetic predispositions, environmental factors, and lifestyle changes, create a sustained demand for reliable diagnostic tools. Technological advancements, particularly in immunoassay techniques, have led to the development of more sensitive, specific, and faster diagnostic assays, improving diagnostic accuracy and enabling earlier disease detection. Growing awareness among healthcare providers and patients regarding the importance of early diagnosis and proactive management of autoimmune conditions also contributes significantly. Furthermore, supportive government initiatives and increasing healthcare expenditure, especially in emerging economies, are expanding access to diagnostic services and driving market expansion. The ongoing shift towards personalized medicine, where diagnostics play a crucial role in tailoring treatment strategies, further underpins market growth.
- Rising Prevalence of Autoimmune Diseases: A global surge in conditions like rheumatoid arthritis, lupus, and inflammatory bowel disease.
- Technological Advancements: Innovations in ELISA, CLIA, and multiplex assay technologies enhancing sensitivity and specificity.
- Increased Healthcare Awareness: Growing recognition of the importance of early diagnosis and treatment initiation.
- Supportive Healthcare Policies: Government investments in diagnostics and public health initiatives.
Obstacles in the Autoimmune Diagnosis Reagents Market
Despite the positive growth trajectory, the Autoimmune Diagnosis Reagents market faces several obstacles. The stringent regulatory approval processes in various regions can be time-consuming and costly, hindering the timely launch of new products. The high cost of advanced diagnostic assays and automated platforms can limit accessibility, particularly in resource-constrained settings. Supply chain disruptions, as witnessed during global health crises, can impact the availability of raw materials and finished products, leading to potential delays and increased costs. Moreover, the market is characterized by intense competition, with established players and emerging companies vying for market share, which can exert pressure on pricing and profit margins. The need for skilled personnel to operate and interpret results from sophisticated diagnostic equipment also presents a challenge in certain regions.
- Regulatory Hurdles: Lengthy and complex approval processes for new diagnostic reagents.
- High Cost of Diagnostics: Limited accessibility in developing economies due to expensive assays and equipment.
- Supply Chain Volatility: Vulnerability to disruptions affecting raw material availability and product delivery.
- Intense Market Competition: Price pressures and the need for continuous innovation to maintain market position.
Future Opportunities in Autoimmune Diagnosis Reagents
The future of the Autoimmune Diagnosis Reagents market is ripe with opportunities. The development of point-of-care testing (POCT) solutions for autoimmune diseases holds immense potential, enabling rapid diagnosis and faster initiation of treatment, particularly in remote or underserved areas. Advances in genomic and proteomic research are uncovering novel biomarkers, creating avenues for the development of new diagnostic tests for currently difficult-to-diagnose autoimmune conditions. The growing trend towards personalized medicine will drive the demand for companion diagnostics that predict patient response to specific therapies. Furthermore, the expansion of healthcare infrastructure and increasing disposable incomes in emerging economies present significant untapped markets for autoimmune diagnostic reagents. The increasing focus on preventive healthcare and early screening programs will also contribute to market growth.
- Point-of-Care Testing (POCT): Development of rapid, portable diagnostic devices for immediate results.
- Novel Biomarker Discovery: Identification of new targets for enhanced diagnostic accuracy and early detection.
- Personalized Medicine: Demand for companion diagnostics to guide targeted therapeutic interventions.
- Emerging Markets: Untapped potential in developing economies with expanding healthcare access.
Major Players in the Autoimmune Diagnosis Reagents Ecosystem
- Thermo Scientific Phadia
- Dr. Fooke Laboratorien
- Siemens Healthineers
- EUROIMMUN
- HOB Biotech
- SDR Diagnostics
- Shenzhen YHLO
- Werfen
- Kexin Biotech
- HUMAN
- AESKU
- Bio-Rad
Key Developments in Autoimmune Diagnosis Reagents Industry
- 2023/08: EUROIMMUN launched a new multiplex assay for autoimmune encephalitis, enhancing diagnostic capabilities for complex neurological disorders.
- 2023/05: Thermo Scientific Phadia expanded its autoimmune diagnostic portfolio with the introduction of novel antibody panels for rare autoimmune conditions.
- 2022/11: Siemens Healthineers announced a strategic partnership with a leading research institute to accelerate the development of advanced autoimmune diagnostic solutions.
- 2022/07: Dr. Fooke Laboratorien introduced an automated platform significantly reducing turnaround times for key autoimmune disease markers.
- 2021/12: Werfen acquired a significant stake in a biotech company specializing in novel autoimmune biomarker discovery.
- 2021/04: HOB Biotech launched a new generation of ELISA kits with improved sensitivity for early detection of celiac disease.
- 2020/10: Bio-Rad introduced a new automated immunoassay system for autoimmune diagnostics, enhancing laboratory efficiency.
- 2020/03: HUMAN expanded its distribution network in Asia-Pacific to increase accessibility of its autoimmune diagnostic reagents.
- 2019/09: AESKU presented research findings on next-generation autoantibody detection methods at a major immunology conference.
- 2019/06: Shenzhen YHLO announced the CE certification for its comprehensive autoimmune diagnostic reagent kit.
Strategic Autoimmune Diagnosis Reagents Market Forecast
The strategic Autoimmune Diagnosis Reagents market forecast indicates robust and sustained growth driven by the confluence of an increasing global burden of autoimmune diseases and continuous technological innovation. The market is poised to benefit from the growing demand for early, accurate, and efficient diagnostic solutions. Key growth catalysts include the development and adoption of highly multiplexed and automated assay platforms, the expansion of point-of-care testing capabilities, and the discovery of novel biomarkers for a wider spectrum of autoimmune conditions. Furthermore, the increasing emphasis on personalized medicine will create opportunities for companion diagnostics, while the expanding healthcare infrastructure in emerging economies presents significant untapped market potential. Strategic collaborations, mergers, and acquisitions are expected to continue shaping the competitive landscape, fostering innovation and expanding market reach, ultimately leading to an enhanced ability to diagnose and manage autoimmune diseases worldwide.
Autoimmune Diagnosis Reagents Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Third-party Testing Agency
-
2. Types
- 2.1. Rheumatology
- 2.2. Hepatology
- 2.3. Gastroenterology
- 2.4. Endocrinology
- 2.5. Others
Autoimmune Diagnosis Reagents Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
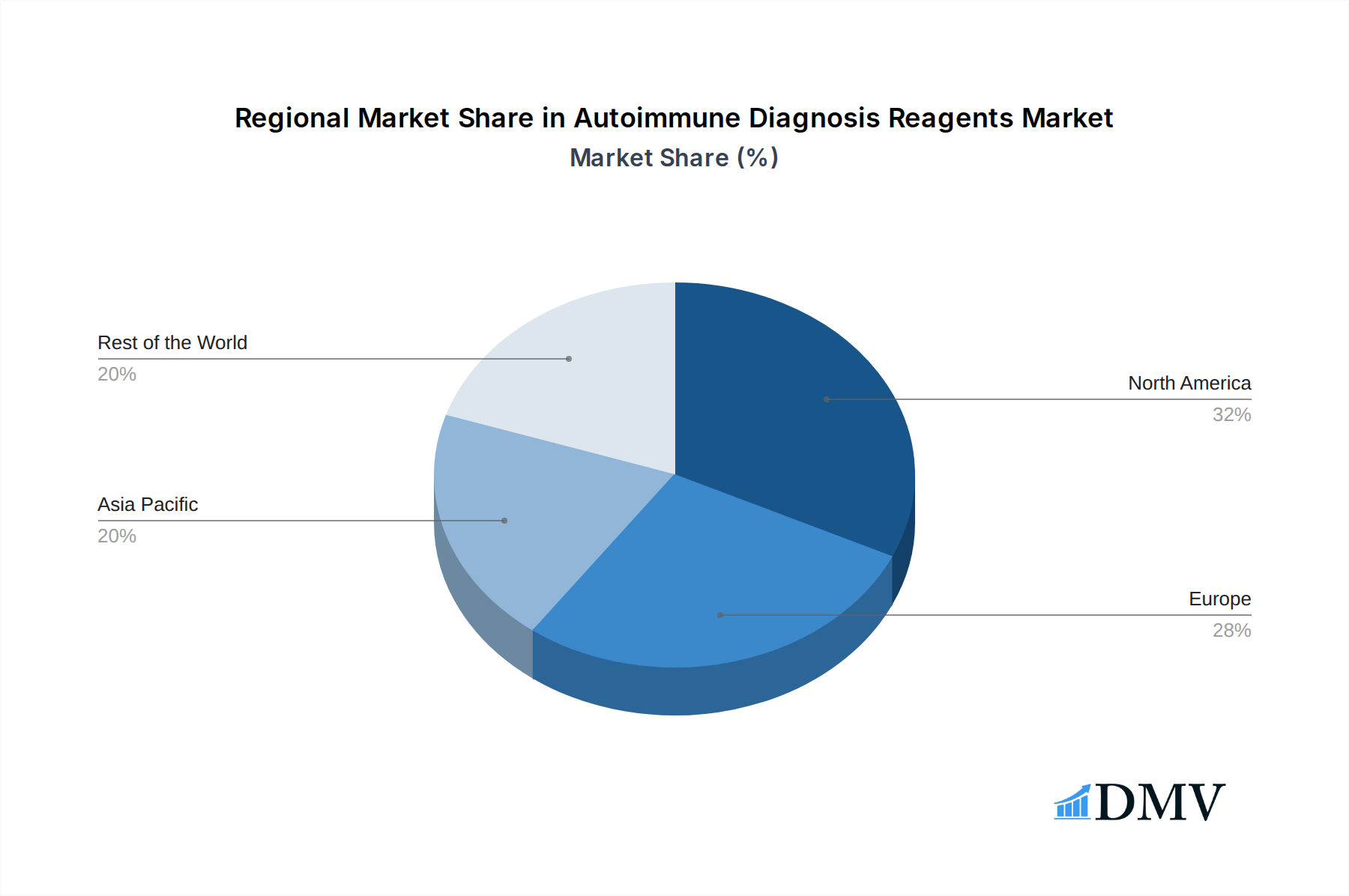
Autoimmune Diagnosis Reagents Regional Market Share

Geographic Coverage of Autoimmune Diagnosis Reagents
Autoimmune Diagnosis Reagents REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.1% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. DMV Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Third-party Testing Agency
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Rheumatology
- 5.2.2. Hepatology
- 5.2.3. Gastroenterology
- 5.2.4. Endocrinology
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Autoimmune Diagnosis Reagents Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Third-party Testing Agency
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Rheumatology
- 6.2.2. Hepatology
- 6.2.3. Gastroenterology
- 6.2.4. Endocrinology
- 6.2.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Autoimmune Diagnosis Reagents Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Third-party Testing Agency
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Rheumatology
- 7.2.2. Hepatology
- 7.2.3. Gastroenterology
- 7.2.4. Endocrinology
- 7.2.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Autoimmune Diagnosis Reagents Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Third-party Testing Agency
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Rheumatology
- 8.2.2. Hepatology
- 8.2.3. Gastroenterology
- 8.2.4. Endocrinology
- 8.2.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Autoimmune Diagnosis Reagents Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Third-party Testing Agency
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Rheumatology
- 9.2.2. Hepatology
- 9.2.3. Gastroenterology
- 9.2.4. Endocrinology
- 9.2.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Autoimmune Diagnosis Reagents Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Third-party Testing Agency
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Rheumatology
- 10.2.2. Hepatology
- 10.2.3. Gastroenterology
- 10.2.4. Endocrinology
- 10.2.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Autoimmune Diagnosis Reagents Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Third-party Testing Agency
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Rheumatology
- 11.2.2. Hepatology
- 11.2.3. Gastroenterology
- 11.2.4. Endocrinology
- 11.2.5. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Thermo Scientific Phadia
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Dr. Fooke Laboratorien
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Siemens Healthineers
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 EUROIMMUN
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 HOB Biotech
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 SDR Diagnostics
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Shenzhen YHLO
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Werfen
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Kexin Biotech
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 HUMAN
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 AESKU
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Bio-Rad
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.1 Thermo Scientific Phadia
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Autoimmune Diagnosis Reagents Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Autoimmune Diagnosis Reagents Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Autoimmune Diagnosis Reagents Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Autoimmune Diagnosis Reagents Volume (K), by Application 2025 & 2033
- Figure 5: North America Autoimmune Diagnosis Reagents Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Autoimmune Diagnosis Reagents Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Autoimmune Diagnosis Reagents Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America Autoimmune Diagnosis Reagents Volume (K), by Types 2025 & 2033
- Figure 9: North America Autoimmune Diagnosis Reagents Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Autoimmune Diagnosis Reagents Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Autoimmune Diagnosis Reagents Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Autoimmune Diagnosis Reagents Volume (K), by Country 2025 & 2033
- Figure 13: North America Autoimmune Diagnosis Reagents Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Autoimmune Diagnosis Reagents Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Autoimmune Diagnosis Reagents Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Autoimmune Diagnosis Reagents Volume (K), by Application 2025 & 2033
- Figure 17: South America Autoimmune Diagnosis Reagents Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Autoimmune Diagnosis Reagents Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Autoimmune Diagnosis Reagents Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America Autoimmune Diagnosis Reagents Volume (K), by Types 2025 & 2033
- Figure 21: South America Autoimmune Diagnosis Reagents Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Autoimmune Diagnosis Reagents Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Autoimmune Diagnosis Reagents Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Autoimmune Diagnosis Reagents Volume (K), by Country 2025 & 2033
- Figure 25: South America Autoimmune Diagnosis Reagents Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Autoimmune Diagnosis Reagents Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Autoimmune Diagnosis Reagents Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Autoimmune Diagnosis Reagents Volume (K), by Application 2025 & 2033
- Figure 29: Europe Autoimmune Diagnosis Reagents Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Autoimmune Diagnosis Reagents Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Autoimmune Diagnosis Reagents Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe Autoimmune Diagnosis Reagents Volume (K), by Types 2025 & 2033
- Figure 33: Europe Autoimmune Diagnosis Reagents Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Autoimmune Diagnosis Reagents Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Autoimmune Diagnosis Reagents Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Autoimmune Diagnosis Reagents Volume (K), by Country 2025 & 2033
- Figure 37: Europe Autoimmune Diagnosis Reagents Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Autoimmune Diagnosis Reagents Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Autoimmune Diagnosis Reagents Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Autoimmune Diagnosis Reagents Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Autoimmune Diagnosis Reagents Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Autoimmune Diagnosis Reagents Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Autoimmune Diagnosis Reagents Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa Autoimmune Diagnosis Reagents Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Autoimmune Diagnosis Reagents Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Autoimmune Diagnosis Reagents Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Autoimmune Diagnosis Reagents Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Autoimmune Diagnosis Reagents Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Autoimmune Diagnosis Reagents Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Autoimmune Diagnosis Reagents Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Autoimmune Diagnosis Reagents Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Autoimmune Diagnosis Reagents Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Autoimmune Diagnosis Reagents Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Autoimmune Diagnosis Reagents Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Autoimmune Diagnosis Reagents Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific Autoimmune Diagnosis Reagents Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Autoimmune Diagnosis Reagents Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Autoimmune Diagnosis Reagents Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Autoimmune Diagnosis Reagents Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Autoimmune Diagnosis Reagents Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Autoimmune Diagnosis Reagents Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Autoimmune Diagnosis Reagents Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Autoimmune Diagnosis Reagents Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Autoimmune Diagnosis Reagents Volume K Forecast, by Country 2020 & 2033
- Table 79: China Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Autoimmune Diagnosis Reagents Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Autoimmune Diagnosis Reagents Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Autoimmune Diagnosis Reagents?
The projected CAGR is approximately 5.1%.
2. Which companies are prominent players in the Autoimmune Diagnosis Reagents?
Key companies in the market include Thermo Scientific Phadia, Dr. Fooke Laboratorien, Siemens Healthineers, EUROIMMUN, HOB Biotech, SDR Diagnostics, Shenzhen YHLO, Werfen, Kexin Biotech, HUMAN, AESKU, Bio-Rad.
3. What are the main segments of the Autoimmune Diagnosis Reagents?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Autoimmune Diagnosis Reagents," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Autoimmune Diagnosis Reagents report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Autoimmune Diagnosis Reagents?
To stay informed about further developments, trends, and reports in the Autoimmune Diagnosis Reagents, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


